Your colleague reports that the conversion to n-butane from the gas phase hydrogenation reaction of 1-butene increases
Question:
Your colleague reports that the conversion to n-butane from the gas phase hydrogenation reaction of 1-butene increases as temperature increases: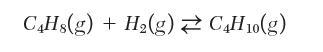
Is this possible? Explain.
Transcribed Image Text:
C4Hs(g) + H(g) C4H0(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Yes it is possible that the conversion of 1butene to nbutane increases as the temperature increases ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
A baseball player usually has four at bats each game. Suppose the baseball player is a lifetime 0.25 hitter. Find the probability that this player will have: (a) Two hits out of four at bats (b) No...
-
Why is the price elasticity of demand a relative measure? That is, why is elasticity measured in percentage terms rather than absolute terms? 1. So the coeffecient of elasticity will not be dependent...
-
Change the given angles to equal angles expressed to the nearest minute. 142.87
-
Prepare journal entries to record the following merchandising transactions of Yang Company, which applies the perpetual inventory system. May 2 Purchased merchandise from Bots Co. for $9,000 under...
-
Tele-Reco is a new specialty store that sells television sets, videotape recorders, video games, and other television-related products. A new Japanese-manufactured videotape recorder costs Tele-Reco...
-
Assume the following for Company C: Revenue growth=5%, CF 0 =5,000, Cash flow growth=2%, NI 0 =6,000, Net Income growth=3%, Discount Rate(WACC)=10%, and COE=14%. Also, CF=Cash Flow, Cash Flow is...
-
Which of the following conditions would you use if you needed to develop an industrial process to produce ethanol from acetylene? Explain. (a) 25C and 1 bar. (b) 250C and 1 bar. (c) 25C and 150 bar....
-
Consider the gas phase hydrogenation reaction of propylene to form propane: To increase the equilibrium conversion, would it help to (a) Increase the pressure? (b) Increase the temperature? (c) Add...
-
_______________ act as facilitators in a market, and they bring together buyers and sellers for a transaction. a. Dealers b. Brokers c. Advisers d. Comptrollers
-
What is the output generated by this method when it is called with a parameter of 3?
-
Explain the primary arguments made by the dissenting opinions in Heller to support their argument that the DC handgun restrictions did not violate the Second Amendment. Your answer should be at least...
-
What is a credit card? What is a debit card? What is a checking account? What is a savings account? What is a money market account?
-
Lean Transportation Principles and Basics of Transportation -Find a product that is manufactured in one country but requires supply from another country. -Determine the value of shipping 1 container...
-
Presented below is information related to Kingbird Company at December 31, 2025, the end of its first year of operations. Sales revenue Cost of goods sold Selling and administrative expenses Gain on...
-
The following is a December 31, 2011, post-closing trial balance for the Jackson Corporation. Required: Prepare a classified balance sheet for Jackson Corporation at December 31, 2011.
-
A summary of changes in Pen Corporation's Investment in Sam account from January 1, 2011, to December 31, 2013, follows (in thousands): ADDITIONAL INFORMATION 1. Pen acquired its 80 percent interest...
-
Name the functional group(s) present in each of the compounds in Problem 2.17.
-
Name the functional group(s) present in each of the compounds in Problem 2.18.
-
Determine whether these structures represent the same compound orisomers: a) b) @ Y il Q0 CHCH3 So d) OCH,
-
@ education.wiley.com [ g [ Question 3 of 3 - Homework: Chapter 2 & Homework: Chapter 2 Question 3 of 3
-
Current Attempt in Progress Wildhorse Company accumulates the following data concerning a mixed cost, using kilometres as the activity level: Kilometres KilometresDriven Total Cost Driven Total...
-
On 30 April 2020, a company classified a freehold building as held for sale. The building had a carrying amount on that date of 5m. The building has been valued by an estate agent at 6m and selling...

Study smarter with the SolutionInn App


