Your coworker has scribbled down the saturation pressures for a pure species from the solid (sublimation) and
Question:
Your coworker has scribbled down the saturation pressures for a pure species from the solid (sublimation) and liquid (evaporation) as follows: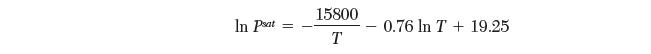
and,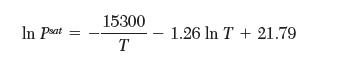
However, in his haste, he forgot to note which equation was for sublimation and which was for evaporation. Please help your coworker by determining the correct matches. Explain your reasoning.
Transcribed Image Text:
In Pat 15800 T 0.76 In T+ 19.25
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Northern Virginia Community College HOW MUCH FINANCIAL RISK SHOULD YOU TAKE? Mark D. D'Antonio Nova Southeastern University FORT LAUDERDALE, FLORIDA, U.S.A. Abstract A successful retirement...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Following procedures similar to those for the homogeneous problem (see Section 8.4.1), develop the following stress field for a pressurized hole in an infinite nonhomogeneous medium with moduli...
-
Regarding government budgets, explain the incrementalism model (championed by Lindblom). How does it change with the level of government? How is it affected by a divided government? Is it an...
-
Calculate the indicated areas. All data are accurate to at least two significant digits. The widths of the baseball playing area in Bostons Fenway Park at 45-ft intervals are shown in Fig. 2.110....
-
The following quotations are available to you. (You may either buy or sell at the stated rates.) Won714.00/S$ HK$4.70/S$ Singapore bank: Singapore dollar quote for Korean won Hong Kong bank: HKS...
-
Martha and Lew are married taxpayers with $400 of foreign tax withholding from dividends in a mutual fund. They have enough foreign income from the mutual fund to claim the full $400 as a foreign tax...
-
Evaluate the definite integral. 5 4 1 5u du (u+5) 5 1 5u du= 4 (u+5) (Type an exact answer in terms of e.
-
(a) A pure fl uid shows the following s vs. T behavior. Draw schematically how the chemical potential would change with temperature. (b) A pure substance shows the following v vs. P behavior at...
-
A well-insulated tank with a valve at the top contains saturated water at 5 MPa. The quality of the water is 0.1. (a) What is the ratio of the liquid volume to the vapor volume? (b) The valve is...
-
Find examples in current news publications similar to those in this chapter of the strategic responses of individual businesses to changes in currency exchange rates. Are these firms adapting to the...
-
Randy and Vonny were coconspirators involved in a drug distribution conspiracy. Randy was caught by police officers and was eager to make a deal that would allow him to remain on the street and act...
-
On 1 January 2013, Xiao and Chen signed a lease contract, under which Xiao let his business premises to Chen as a sports store for five years, with a yearly rental of RMB 600,000 payable on the last...
-
Textra produces parts for a machine manufacturer. Parts go through two departments, Molding and Trimming. The company budgets overhead cost of $240,000 in the Molding department and $200,000 in the...
-
2-facility requires lifting heavy boxes from a pallet. The following posture is observed where the torso flexion is 30 (i.e., assume a= 30) in lifting a box. Assume this is a symmetrical lift where...
-
What is an electron volt (eV)? Demonstrate that 1.0000u of mass is the equivalent of 931.5MeV of energy, using Einstein's equation. Use precise values to ensure a precise answer. Be aware the answer...
-
On December 31, 2011, Interlink Communications issued 6% stated rate bonds with a face amount of $100 million. The bonds mature on December 31, 2041. Interest is payable annually on each December 31,...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Treat carbon monoxide as a perfect gas and apply equilibrium statistical thermodynamics to the study of its properties, as specified below, in the temperature range 100-1000 K at 1 bar. V = 2169.8...
-
The exchange of deuterium between acid and water is an important type of equilibrium, and we can examine it using spectroscopic data on the molecules. Calculate the equilibrium constant at (a) 298 K...
-
Suppose that an intermolecular potential has a hard-sphere core of radius 'I and a shallow attractive well of uniform depth E out to a distance '2' Show, by using eqn 17.42 and the condition E kT,...
-
Nancy is 21 and a full-time college student and is blind. Nancy lives with her mother and is claimed as a dependent by her mother. Her earned income was $6,500 for the 2022 tax year. Using the...
-
Christopher Foley, an attorney, has a law corporation, Christopher Foley, Attorney, Inc., that began the year with total assets of $145,000, total liabilities of $70,000, and stockholders' equity of...
-
Which of the following statements is correct? Group of answer choices All of these answers are correct. A subscriber must take reasonable steps to offer users a receipt for all transactions, before...

Study smarter with the SolutionInn App


