Figure 4-26 shows an energy loss spectrum for He measured in an apparatus such as that shown
Question:
Figure 4-26 shows an energy loss spectrum for He measured in an apparatus such as that shown in Figure 4-24a. Use the spectrum to construct and draw carefully to scale an energy-level diagram for He.
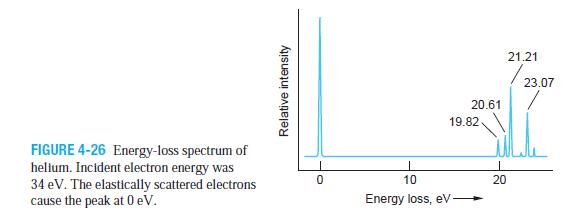
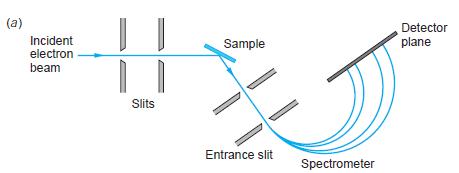
Figure 4-24(a)
Transcribed Image Text:
FIGURE 4-26 Energy-loss spectrum of helium. Incident electron energy was 34 eV. The elastically scattered electrons cause the peak at 0 eV. Relative intensity 0 20.61 19.82 10 Energy loss, eV 20 21.21 23.07
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
23 22 21 20 ...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
An air wedge such as that shown in Fig. 24.30 can be used to measure small dimensions, such as the diameter of a thin wire. (a) If the top glass plate is illuminated with monochromatic light, the...
-
Use the data to construct a 95% confidence interval estimate of the number of credit hours taken by the population of full-time students. Write a brief statement that interprets the result. The...
-
For a finite slope such as that shown in Figure 16.10, assume that the slope failure would occur along a plane (Culmanns assumption). Find the height of the slope for critical equilibrium. Given: ' =...
-
Find the derivative of the function. y = e x-4
-
A study by researchers at the University of Maryland addressed the question of whether the mean body temperature of humans is 98.6F. The results of the study by P. Mackowiak et al. appeared in the...
-
The graph of x 2 /4 - y 2 = 1 is a hyperbola. We know that the graph of this hyperbola approaches its asymptotes as 0 x 0 increases without bound. Work Exercises in order, to see the relationship...
-
Halves Vhidd is a production manager at a metal fabricating plant. Last night he read an article about a new piece of equipment that would dramatically reduce his divisions costs. Harvey was very...
-
What criteria must be met before donated services can be recorded as contribution revenue and an expense? Give an example of a service that might qualify as a donated service for accounting purposes.
-
Given the vector <6, -12>. | <6, -12>|= with a directional angle of
-
correlating branch predictor uses the behavior of the most recent m executed branches to choose from 2m predictors, each of which is an nbit predictor. A two-level local predictor works in a similar...
-
A sample of hydrogen atoms are all in the n = 5 state. If all the atoms return to the ground state, how many different photon energies will be emitted, assuming all possible transitions occur? If...
-
In a sample that contains hydrogen, among other things, four spectral lines are found in the infrared with wavelengths 7460 nm, 4654 nm, 4103 nm, and 3741 nm. Which one does not belong to a hydrogen...
-
The following pedigree shows the inheritance of a dominant trait. What is the chance that the offspring of the following matings will show the trait: (a) III-1 Ã III-3; (b) III-2 Ã...
-
Explain the frequency response of the differential-mode voltage gain.
-
Which of the following is required for a firm to designate itself Member of the American Institute of Certified Public Accountants on its letterhead? a. At least one of the partners must be a member....
-
The CPA should not undertake an engagement that requires independence if his or her fee is to be based on a. The findings of a tax authority. b. A percentage of audited net income. c. Per diem rates...
-
What publication provides guidance to auditors who perform audits of government entities?
-
Describe the series and shunt output connections of a feedback amplifier.
-
Define the market process, the command process, and the traditional process. How does each process deal with the basic questions of what, how, and for whom?
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Two wires are tied to the 300 g sphere shown in FIGURE P8.46. The sphere revolves in a horizontal circle at a constant speed of 7.5 m/s. What is the tension in each of the wires? FIGURE P8.46 1.0 m...
-
Two wires are tied to the 300 g sphere shown in FIGURE P8.46. The sphere revolves in a horizontal circle at a constant speed of 7.5 m/s. What is the tension in each of the wires? FIGURE P8.46 1.0 m...
-
Two wires are tied to the 2.0 kg sphere shown in FIGURE P8.45. The sphere revolves in a horizontal circle at constant speed. a. For what speed is the tension the same in both wires?b. What is the...
-
Question 4 of 6 < > -/15 Current Attempt in Progress Sheridan Company has accounts receivable of $202,000 at September 30, 2024. An analysis of the accounts shows the following: Month of Sale Balance...
-
The charge on your rent-to-own washer is $40 per month. After 32 months, you get to keep the washer. How much does the washing machine end up costing you? Cost of washing machine
-
Prepare horizontal analysis on income statement below: Bristol Myers Squibb Co. (NYS: BMY) Report Date 2020 2019 2018 Net product sales 41,321 25,174 21,581 Alliance & other revenues 1,197 971 980...

Study smarter with the SolutionInn App


