According to Equation 9.30, the vibrationrotation constant for a harmonic oscillator is not zero and is in
Question:
According to Equation 9.30, the vibration–rotation constant for a harmonic oscillator is not zero and is in fact negative. Consider the rotational constant for the v = 0 state of a harmonic oscillator, which is proportional to the expectation value {1/R}V = 0. Show that this expectation value is greater than 1/R2e, thus B0 for a harmonic oscillator is greater than Be, resulting in a negative vibration–rotation coupling constant.
Equation 9.30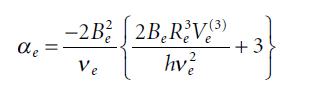
Transcribed Image Text:
de -2B²2B₂RV(3) 2B, R/V + 3} +3 hv² Ve
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The equation provided represents the vibrationrotation interaction constant a for a diatomic molecul...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
According to quantum mechanics, the energy of a harmonic oscillator is quantized. That is, it can take on any one of a certain set of values, given by where h is Plancks constant, equal to 6.62608...
-
For a harmonic oscillator of effective mass 2.88 x 10-25kg, the difference in adjacent energy levels is 3.17 z calculate the force constant of the oscillator.
-
The ground-state energy of a harmonic oscillator is 5.60eV. If the oscillator undergoes a transition from its n = 3 to n = 2 level by emitting a photon, what is the wavelength of the photon?
-
Dr. Bold has a personal automobile policy with liability limits as follows: $100,000/$300,000 BI and $50,000 PD. Dr. Bold is held liable in an accident in which he must pay for bodily injuries as...
-
Zurg Corp. uses the direct method to prepare its statement of cash flows. Zurgs trial balances at December 31, 2014 and 2013 are as follows. Additional information: 1. Zurg purchased a major piece of...
-
Three Exchange Rates are as follows: 1) US Dollars (USD) to Japanese Yens (JYP) at JPY 95 to USD1 2) JPY to Euros (EUR) at JPY 124 to EUR 1 3) EUR to USD at EUR 0.72 to USD 1 Suppose you start with...
-
6. MONEY Campaign contributions and spending are largely uncontrolled.
-
On May 1, Arnie Watson sent a memo to his boss, the director of project management, stating that the MX project would require thirteen weeks for completion according to the figure shown at the top of...
-
urrent Attempt in Progress Cullumber Industries purchased the following assets and constructed a building as well. All this was done during the current year. Assets 1 and 2: These assets were...
-
Use the data in Table 9.2 to assign the Q branch lines in Figure 9.6. Account for the relative intensities. Figure 9.6 Table 9.2 Spectroscopic constants for diatomics in the ground electronic state...
-
Use the harmonic oscillator raising and lowering operators to derive the selection rules for vibrational transitions that result from electrical anharmonicity in the form (/dq)oq.
-
Junkers Stash started the 2016 accounting period with the balances given in the financial statements model shown below. During 2016 Junkers Stash experienced the following business events: 1. Paid...
-
The pharmaceutical company is in the planning stages of a critical project and the project management team is finalizing the responsibility assignment matrix. How will this document benefit the team?...
-
The customer has just attended the weekly staff meeting for the infrastructure project and has stated that the market is changing for the product being created. As a result of this, the customer has...
-
Of the following, which is the most comprehensive definition of a project? a. An environment created to deliver a product, service, or result. b. A collaborative enterprise that delivers a product,...
-
A new call center is being built to support a new product at a national telephone company. The company doesnt have any data on how long it will take to sign up customers via the call center. This...
-
There is $150,000 allocated for management reserves. One of the stakeholders is a newly hired vice president who comes from a small company where project management was not formalized, so he is...
-
Why does the existence of a potent Pigou Effect guarantee a negatively sloped AD curve?
-
In a nonmagnetic medium, E = 50 cos (10 9 t 8x) a y + 40 sin (10 9 t 8x) a z V/m find the dielectric constant r and the corresponding H.
-
How many constitutional isomers are obtained when each of the following compounds undergoes monochlorination? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j)
-
Propylene is produced by cracking petroleum and is a very useful precursor in the production of many useful polymers. Propylene has one constitutional isomer. Draw that isomer, and identify its...
-
Identify the reagents you would use to achieve each of the following transformations: (a) (b) (c) (d) (e) OTs `CN
-
Se utiliza suavizacin exponencial simple para pronosticar lademanda. El pronstico anterior de 66 result ser cinco unidadesmenos que la demanda real. El siguiente pronstico es 67,5, lo queim 2 answers
-
Q7Q9 Exercise 7-37 (Algo) Prorate Over- or Underapplied Overhead (LO 7-3) Marian Manufacturing (2M) applies manufacturing overhead to jobs based on direct labor costs. For Year 2, 2M estimates its...
-
The Adam Manufacturing Company in Rochester, Minnesota, assembles and tests electronic components used in smartphones. Consider the following data regarding component T24 (amounts are per unit)...

Study smarter with the SolutionInn App


