Assign the bands in the benzene spectra displayed in Figures 10.12 and 10.13. Figure 10.12 Figure 10.13
Question:
Assign the bands in the benzene spectra displayed in Figures 10.12 and 10.13.
Figure 10.12
Figure 10.13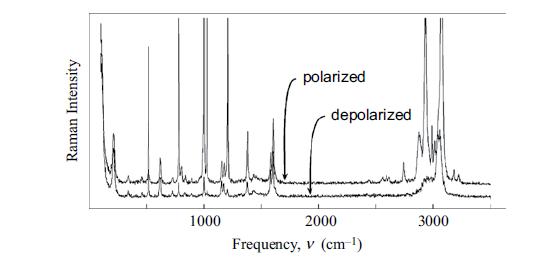
Transcribed Image Text:
Absorbance benzene Qu m. 2000 Frequency, v (cm-¹) toluene 1000 3000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
The provided images show the spectra of benzene indicating various absorption and Raman intensity ba...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The infrared spectra for three compounds are provided. Each compound has one or more of the following functional groups: conjugated ketone, ester, amide, nitrile, and alkyne. Determine the functional...
-
Spectra are given for three compounds. Each compound has one or more of the following functional groups: alcohol, amine, ketone, aldehyde, and carboxylic acid. Determine the functional group(s) in...
-
The following spectra are taken from a compound that is an important starting material for organic synthesis. Determine the structure, first by considering each spectrum individually, then by...
-
Adam Hoover bought 72 shares of General Oil Co. stock at a par value of $85 per share. The stock paid annual dividends of 7 1/2%. How much did Adam receive in dividends this year? Commissions 0. Mrs....
-
Ryan Inc. had the following condensed balance sheet at the end of operations for 2013. During 2014 the following occurred. 1. Land was purchased for $29,000. 2. Notes payable in the amount of $45,500...
-
Question 9In the past five years, Eagle Burger has grown to over 200stores. Two of the company-operated units, Northside and Southside,are among the fastest-growing stores.Both are considering exp 2...
-
4. Federal antitrust statutes are complex, but the basic goal is straightforward: to prevent a major industry from being so dominated by a small group of corporations that they destroy competition...
-
Martin Buber Co. purchased land as a factory site for $400,000. The process of tearing down two old buildings on the site and constructing the factory required 6 months. The company paid $42,000 to...
-
Iridescent Corporation manufactures and sells two types of decorative lamps, Knox and Ayer. It expects to manufacture 18,000 Knox lamps and 9,000 Ayer lamps in 2017. The following data are available...
-
The absorption spectrum of I 2 has E 00 /hc = 15,677 cm 1 , and the onset of the continuum is at 19,735 cm 1 . The excited state dissociates to I( 2 P 3/2 ) + I( 2 P 1/2 ), which is 7589 cm 1 above...
-
The SO 2 molecule, analogous to H 2 O, has a 1 A 1 ground electronic state with the valence electronic configuration (1a 2 ) 2 (4b 2 ) 2 (6a 1 ) 2 (2b 1 ) 0 and low-lying excited singlet states of...
-
If someone conducted one-way ANOVAs predicting the clustering variables using the cluster membership variable that was obtained from a cluster analysis procedure, what should the expectation of the...
-
What is Zeroth law of thermodynamics.
-
All of the following interpersonal or soft skills are applicable to managing stakeholder engagement except . . . a. Building trust b. Minimizing conflict c. Active listening d. Overcoming resistance...
-
What are the three categories of processing control?
-
Explain four advantages of NC machine.
-
What are some typical problems with passwords?
-
Explain the importance of the assumption of fixed nominal wages in the determination of the short-run aggregate supply curve.
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
Identify all products expected for each of the following reactions. Take stereochemistry into account, and draw expected stereoisomer(s), if any: (a) (b) (c) (d) NBS hv
-
Ambien TM is a sedative used in the treatment of insomnia. It was discovered in 1982 and brought to market in 1992 (it takes a long time for new drugs to undergo the extensive testing required to...
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Question Content AreaBond premium, entries for bonds payable transactions, interestmethod of amortizing bond premiumRodgers Corporation produces and sells football equipment. OnJuly 1, 20Y1, Rodge 2...
-
Communication can be described as the and exchange of information. 000 phasing preparing transfer talking
-
Q7Q5 Fuiton Construction is a general contractor for large construction projects. The budget costs and the time to reach a particular percentage of completion (in months) follows. Also shown are the...

Study smarter with the SolutionInn App


