The ground electronic state of Cr 3+ in an octahedral field is 4 A 2 g ,
Question:
The ground electronic state of Cr3+ in an octahedral field is 4A2g, which derives from a 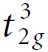 configuration. Is this state subject to Jahn–Teller distortion?
configuration. Is this state subject to Jahn–Teller distortion?
Transcribed Image Text:
3 28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The JahnTeller theorem predicts that a nonlinear molecule or a nonlinear complex ion in a degenerate ...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The peak in the UV-VIS spectrum of acetone [(CH3)2CO] corresponding to the n * transition appears at 279 nm when hexane is the solvent, but shifts to 262 nm in water. Which is more polar, the ground...
-
The peak in the UV-VIS spectrum of acetone [(CH3)2CoeO] corresponding to the n * transition appears at 279 nm when hexane is the solvent, but shifts to 262 nm in water. Which is more polar, the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
EG Corporation redeemed 200 shares of stock from one of its shareholders in exchange for $200,000. The redemption represented 20% of the corporation's outstanding stock. The redemption was treated as...
-
Presented below are data taken from the records of Natural Homes Company. Additional information: 1. Held-to-maturity securities carried at a cost of $25,000 on December 31, 2013, were sold in 2014...
-
Worksheet 10.1 DS 123 Name Suppose women's athletic shoes have a mean price of $75.15 and a standard deviation of $17.89. What is the probability that the mean price of a random sample of 50 pairs of...
-
2. YOU BE THE JUDGE WRITING PROBLEM An off-duty, out-of-uniform police officer and his son purchased some food from a 7-Eleven store and were still in the parking lot when a carload of teenagers...
-
Lower-of-Cost-or-Market Sedato Company follows the practice of pricing its inventory at the lower-of-cost-or-market, on an individual-item basis. From the information above, determine the amount of...
-
If Toyota Motor Company receives an order on May 1, begins production on May 22, and ships the order on May 30 immediately following production, then what is the manufacturing cycle efficiency (MCE)...
-
The SO 2 molecule, analogous to H 2 O, has a 1 A 1 ground electronic state with the valence electronic configuration (1a 2 ) 2 (4b 2 ) 2 (6a 1 ) 2 (2b 1 ) 0 and low-lying excited singlet states of...
-
The compound shown below has ma x = 736 nm in hexane and 553 nm in water. Explain the basis for the solvent shift. H3C H3C Z NO
-
What are the primary goals of a court in interpreting a contract?
-
Define refrigeration and air conditioning.
-
The team is in the process of completing the work breakdown structure of the project. Once they have completed this, which of the following can they begin to work on? a. Estimating costs b....
-
Payroll is often used as a good example of batch processing using sequential files. Explain why.
-
Define internal energy and enthalpy.
-
The project manager always involves the team in the creation of the work breakdown structure. What is the most significant benefit derived from this approach? a. Generation of a more accurate...
-
Discuss what caused aggregate demand to collapse during the Great Depression and whether any of those were similar to why aggregate demand declined during the Global Economic Crisis.
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
Each of the following molecules has one plane of symmetry. Find the plane of symmetry in each case: a. b. c. d. e. f. CI Me Me Br
-
Compound A has molecular formula C5H12, and monobromination of compound A produces only compound B. When compound B is treated with a strong base, a mixture is obtained containing compound C and...
-
Compare the Lewis dot structure of nitrogen and phosphorus and explain why you might expect these two atoms to exhibit similar bonding properties?
-
Sadie gave you check number 6877 for $75 from Sundown Dog Rescue. Sadie had agreed to give dogs an outdoor bath for $15; for each bath, the rescue kept $10 and gave Sadie $5. Along with the check,...
-
Im having an issue with implementing LCFS with my code, I need help figuring out what Im doing wrong. Thank you in advance!! This is all being done in DelivC fyi Here's the assignment prompt: ...
-
Tott Company has the following internal control proceduresover cash disbursements. Identify the internal control principethat is applicable to each procedure. a. b. C. d. e. Company checksare prenu...

Study smarter with the SolutionInn App


