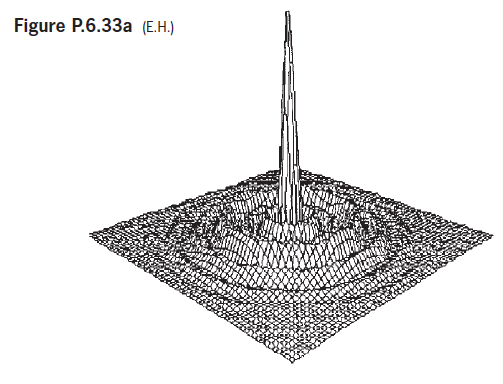
Figure P.6.33 shows the image irradiance distributions arising when a monochromatic point source illuminates three different optical
Question:

Transcribed Image Text:
Figure P.6.33a (ЕН) Figure P.6.33b (ЕН) Figure P.6.33c (EН)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Because a it is symmetrical and looks like a somewhat alter...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Figure P.10.30 is a computer-generated Fraunhofer irradiance distribution. Describe the aperture that would give rise to such a pattern and give your reasoning in detail. Figure P.10.30
-
Figure shows cross sections through two large, parallel, non-conducting sheets with identical distributions of positive charge with surface charge density = 1.77 x 10-22C/m2. In unit-vector...
-
Two monochromatic point sources radiate in-phase. At the usual distant plane of observation (parallel to the line connecting the sources) the irradiance from one of them is 100 times the irradiance...
-
Prepare journal entries to record each of the following sales transactions for the sales company. The company uses a perpetual inventory system and the gross method. 1. April 1st Sold for $3,000,...
-
On January 1, 2017, Abbey acquires 90 percent of Benjamin's outstanding shares. Financial information for these two companies for the years 2017 and 2018 follows: Assume that a tax rate of 35 percent...
-
In New York City, 2406-12 Amsterdam Associates, LLC, brought an action in a New York state court against Alianza Dominicana and Alianza, LLC, to recover unpaid rent. The plaintiff asserted cause to...
-
Puerto International has a branch in Mexico that manufactures a garage door alarm for people with mountain bike racks that fit on the top of their cars. The subsidiary earned 800,000 dollars in 2000...
-
In The Effect of Nozzle Design on the Stability and Performance of Turbulent Water Jets (Fire Safety Journal, Vol. 4, August 1981), C. Theobald describes an experiment in which a shape measurement...
-
Several studies showed that after eating a very large meal, subjects had increased risk of heart attack. A diet food manufacturer financed this research. Identify What wrong. Choose the correct...
-
4. Sleekfon and Sturdyfon are two major cell phone manufacturers that have recently merged. Their current market sizes are as shown in Table 5-9. All demand is in millions of units. Sleekfon has...
-
Starting with the exact expression given by Eq. (5.5), show that Eq. (6.46) results, rather than Eq. (5.8), when the approximations for 0 and i are improved a bit. 1 ( n2Si R l; NSo ) (5.5) li lo
-
Explain how SQL Server 2014 produces XML output using the FOR XML RAW clause.
-
What are the criteria for price-setter companies?
-
To determine whether merchandise is included in ending inventory, an auditor could test: a. Open purchase orders. b. Purchase cutoff. c. Commitments made by Purchasing. d. Invoices received on or...
-
How can management control against unauthorized or duplicate cash payments?
-
Which of the following financial statement assertions are addressed by the physical observation of inventory counts? a. Existence and valuation. b. Rights and completeness. c. Presentation and...
-
Identify the major business functions and activities common to the expenditure/ disbursement cycle.
-
Which portfolio of debt and equity securities is always reported as a current asset in the balance sheet? a. Treasury shares. b. Held-to-maturity securities. c. Available-for-sale securities. d....
-
The distance d (in meters) of the bob of a pendulum of mass m (in kilograms) from its rest position at time t (in seconds) is given. The bob is released from the left of its rest position and...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Neon-23 beta decays in the following way: 23- 23- NeNa+ e + V
-
Uranium-238 is radioactive and decays into a succession of different elements. The following particles are emitted before the nucleus reaches a stable form: , , , , , , , , , , , , , and ( stands...
-
How much energy must a bombarding proton possess to cause the reaction 7 Li(p,n) 7 Be? Give your answer to three significant figures.
-
The records of Hoffman Company reflected the following balances in the stockholders equity accounts at December 31, 2018: Common stock, par $12 per share, 40,000 shares outstanding. Preferred stock,...
-
Problem 9-12 Direct Materials and Direct Labor Budgets [LO4, LO5] The production department of Prison Company has submitted the following forecast of units to be produced by quarter for the upcoming...
-
Exercise 10-6 (Algo) Contrasting Return on Investment (ROI) and Residual Income [LO10-1, LO10-2] Meiji Isetan Corporation of Japan has two regional divisions with headquarters in Osaka and Yokohama....

Study smarter with the SolutionInn App


