For each of the following complexes, determine the oxidation state of the metal and the total number
Question:
For each of the following complexes, determine the oxidation state of the metal and the total number of valence electrons it possesses.
(a)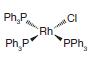
(b)
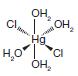
(c)
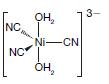
Transcribed Image Text:
Ph.P H Ph P CI PPh mee Rh
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Answer a The oxidation state of the metal in the complex is 3 a...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
For each of the following complexes, determine the oxidation state of the transition-metal atom. a. [CoCl(en)2(NO2)]NO2 b. [PtCl4]2 c. K3[Cr(CN)6] d. [Fe(H2O)5(OH)]2+
-
State the research problems for each of the following management decision problems. a. Should a new product be introduced? b. Should an advertising campaign that has run for three years be changed?...
-
State the appropriate nonparametric test for each of the following examples. (a) A physiological psychologist compares differences in the amount of chemical activity in a certain brain region between...
-
1. Prepare a schedule of cost of goods manufactured for Denim Bones for the year ended December 31, 2024. 2. Prepare an income statement for Denim Bones for the year ended December 31, 2024. 3. How...
-
The percentages of Americans who are satisfied with the way things are in the United States are shown in Table 59 for various years. Let p be the percentage of Americans at t years since 1990 who are...
-
Computing Bad Debts and Preparing Journal Entries the trial balance before adjustment of Estefan Inc. shows the following balances. Give the entry for estimated bad debts assuming that the allowance...
-
The constants obtained by Shende and Sharma (1974) for use in Eq. (16-72) are given in the following table. Assume their experiments with \(\mathrm{NaOH}-\mathrm{SO}_{2}\) were done at \(1.0...
-
AM Express Inc. is considering the purchase of an additional delivery vehicle for $ 55,000 on January 1, 2014. The truck is expected to have a five-year life with an expected residual value of $...
-
3. WB Inc. is funded by common & preferred equity and long-term debt and is subject to a 21% tax rate. Its common equity comprises 40% of its capital structure, while its preferred equity makes up...
-
8.a A Spartan missile and a Trojan missle are both flying at the s ame constant altitude. At time t, the Spartan missile is at the point: Clear[spartan, t] spartan[t_] = {16.1-7t+ t, 13 t - 2 t}...
-
What is the product of the following SuzukiMiyaura coupling? Br B(OR)2 + Pd catalyst Base
-
In The Chemistry of Complex Cross Couplings we showed a Heck reaction used in the synthesis of ecteinascidin 743, a natural compound with anticancer activity isolated from the marine tunicate...
-
Resort Inns, Inc., has a total of 2,000 rooms in its nationwide chain of motels. On average, 70% of the rooms are occupied each day. The companys operating costs are $21 per occupied room per day at...
-
Howe Corporation calculates inventory and cost of goods sold one time at the end of every accounting period. In contrast, Kelty Industries updates their inventory and cost of goods sold accounts...
-
Suppose Investigator Lockemup suspects drug activity at a residence. He can walk to the front door of the residence and knock. There is a caveat that the front door must be accessible and not...
-
Convert the following LP into the standard form: max z = 2x15x23x3 s.t. x12x2 + x3 > 20 2x14x2 x3 = = 50 3x1+x372 x1, x20 x3 free
-
It is a common perception that macOS usersfeel safe from viruses and malware compared to Windows users. Conduct a web search to research the validity of this perception and discuss the steps should...
-
You have a credit card with a balance of $14,500 and an APR of 18.3 percent compounded monthly. You have been making monthly payments of $275 per month, but you have received a substantial raise and...
-
Prepare journal entries to record the following merchandising transactions of Menards, which applies the perpetual inventory system and gross method. July 3 Purchased merchandise from OLB Corp. for...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
Predict the positions of the major absorption bands in the IR spectra of thesecompounds: CH30 CH-NH, a) CH3CH=CHCCH; c) b) C-H NO, e) CH-CH,C3- d) CH,CH,CH,OH CH3
-
Explain how IRD spectroscopy could be used to distinguish between these compounds: CH;CH,C=CH a) CH,CH,CH=CH; and CH3 H- b) CH, nd CH3 CCH3 CH c) and d) CH.CH,CH,C,NH, and CH.CH,NHCH,CH,
-
List the positions of the important absorption bands in the IR spectra of these compounds: b) H,H-CH-C3DN ) -H,H,NH> CCH,CH3 c) CH;CH,COCH;CH,CH3 d)
-
What is the crossover rate for the following two projects? 13.48% 23.21% 23.81% 0.60% 12.26% Year Project X Project Y 0 -$541,200 -$511,500 1 219,600 209,300 2 229,500 219,100 3 236,700 227,000 4...
-
3 Growing Perpetuity (and selling price) Carrie grandfather owns a farm. The farm earned $410,000 this year. The profits are expected to grow at 5.5% per year forever. What is the value of the farm...
-
The beta of four stocks-G, H, I, and J-are 0.43, 0.76, 1.07, and 1,59, respectively. What is the beta of a portfolio with the following weights in each asset: E ortfolio 1?

Study smarter with the SolutionInn App


