Provide a mechanism for the following reaction. H2SO4, H2O OH
Question:
Provide a mechanism for the following reaction.
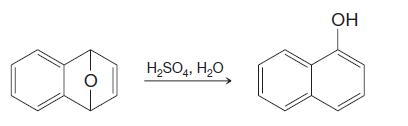
Transcribed Image Text:
H2SO4, H2O OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Answer The reaction shown can be achieved using a Heck reaction In a Heck reac...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Provide a mechanism for the following reaction and explain why it occurs faster than nitration of benzene. NO2
-
Provide a mechanism for the following reaction OH H2SO4 H20
-
Provide a mechanism for the following reaction. Draw a reaction energy coordinate diagram that illustrates the kinetic and thermodynamic pathways for this reaction. HBr Br+
-
The membrane filter technique is used to test a polluted water sample for coliform group. Three different dilutions of the water sample were prepared and each was filtered through 5 filter membranes....
-
X + 5 9 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Suppose that an antique jewelry dealer is interested in purchasing a gold necklace for which the probabilities are 0.22, 0.36, 0.28, and 0.14 respectively, that she will be able to sell it, for a...
-
The stockholders' equity LOS, 6,8 accounts of Cooper Corporation at January 1 follow: During the year, the following transactions occurred: Jan. 5 Issued 20,000 shares of common stock for \(\$ 15\)...
-
MVP Corp uses LIFO to value its inventory. The 20x8 inventory records disclose the following: At December 26, 20x8, the company had a special, nonrecurring opportunity to purchase 40,000 units at $17...
-
You have just invested in a portfolio of three stocks. The amount of money that you invested in each stock and its beta are summarized below. Stock Investment Beta A $218,000 1.50 B 327,000 0.60 C...
-
The graph in Figure 13 shows the amount of water in a city water tank during one day when no water was pumped into the tank. What was the average rate of water usage during the day? How fast was...
-
Starting with aniline, outline a synthesis of each of the following: (a) p-Bromoaniline (b) o-Bromoaniline (c) 2-Bromo-4-nitroaniline (d) 4-Bromo-2-nitroaniline
-
Starting with toluene, outline a synthesis of each of the following: (a) m-Chlorobenzoic acid (b) p-Methylacetophenone (c) 2-Bromo-4-nitrotoluene (d) p-Bromobenzoic acid (e)...
-
When are partnerships mandated to adjust the basis of their assets (inside basis) when a partner sells a partnership interest or receives a partnership distribution?
-
a . If the town decided to use Lindahl pricing ( a tax based on a Lindahl equilibrium ) to collect enough money to provide the socially efficient quantity of acres, what would be each person s tax...
-
Answer the following questions about the forces in the nucleus. 1. What are the two forces in the nucleus? 2. Which one brings stability to the nucleus? 3. Which one brings instability to the...
-
1. Explain and discuss the advantages of nuclear energy technology, considering both fission and fusion. 2. Explain and discuss the disadvantages of nuclear energy technology, considering both...
-
Figure (a) shows, in cross section, two long, parallel wires carrying current and separated by distance L. The ratio i/i2 of their currents is 5.60; the directions of the currents are not indicated....
-
4. The membrane that surrounds a certain type of living cell has a surface area of 5.20 x 10-9 m2 and a thickness of 1.50 x 10-8 m. Assume that the membrane behaves like a parallel plate capacitor...
-
Rolston Music Company is considering the sale of a new sound board used in recording studios. The new board would sell for $26,400, and the company expects to sell 1,500 per year. The company...
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
Explain why only one of the two chlorines of 1, 2-dichloro-2-methylpropane is replaced by a hydroxy group when the compound is heated in water (see the preceding hydrolysis reaction.
-
On the basis of the bond cleavage shown for this reaction in Figure 10.1, predict the stereo chemistry of the product.Explain. OCCH, CH,CH, ." -
-
Show the products of thesereactions: CI CH3CO, NaOH a) DMSO . Br CH,CO, b) DMF CH
-
Caspian Sea Drinks is considering the purchase of a new water filtration system produced by Rube Goldberg Machines. This new equipment, the RGM - 7 0 0 0 , will allow Caspian Sea Drinks to expand...
-
RCD sells cars in R-land, where the currency is the R$. At the start of 20X2, RTW introduced more favourable credit terms and, as a result, credit sales have increased from R$800,000 in 20X1 to...
-
1.Does the organization have qualified employees to assess business income tax? 2.Do the annual plan prepared by the sub city match with the actual performance? 3.Do tax payers maintain book of...

Study smarter with the SolutionInn App


