Show how the diketone at the right could be prepared by a condensation reaction: lion to bolo
Question:
Show how the diketone at the right could be prepared by a condensation reaction:
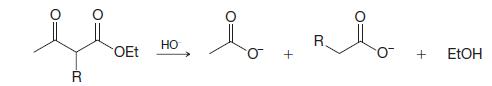
Transcribed Image Text:
lion to bolo HO R OEt + + R EtOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The d ik et one at the right could be prepared by a cond ensation reaction between two molecules of a lde hyde and two molecules of ket one The reacti...View the full answer

Answered By

DHRUV RAI
As a tutor, I have a strong hands-on experience in providing individualized instruction and support to students of all ages and ability levels. I have worked with students in both one-on-one and group settings, and I am skilled in creating engaging and effective lesson plans that meet the unique needs of each student.
I am proficient in using a variety of teaching techniques and approaches, including problem-based learning, inquiry-based learning, and project-based learning. I also have experience in using technology, such as online learning platforms and educational software, to enhance the learning experience for my students.
In addition to my teaching experience, I have also completed advanced coursework in the subjects that I tutor, including mathematics, science, and language arts. This has allowed me to stay up-to-date on the latest educational trends and best practices, and to provide my students with the most current and effective teaching methods.
Overall, my hands-on experience and proficiency as a tutor have equipped me with the knowledge, skills, and expertise to help students achieve their academic goals and succeed in their studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Show how this diketone could be prepared by a condensation reaction:
-
The valuable polymer polyurethane is made by a condensation reaction of alcohols (ROH) with compounds that contain an isocyanate group (RNCO). Two reactions that can generate a urethane monomer are...
-
Show how 2-butanone could be prepared by a procedure in which all of the carbons originate in acetic acid (CH3CO2H).
-
Given an array, Arr[] of integer numbers have size N. The task is to find out single or consecutive numbers from Arr[] with 0 sum. When found, print "1" otherwise "0". Example-1: Input: 4 (6,-2,3,-1)...
-
4x 8 Describe the solution set as an inequality, in interval notation, and on a graph.
-
The following data represent the length of life in years, measured to the nearest tenth, of 39 similar fuel pumps:(a) Construct a stem-and-leaf plot for the life in years of the fuel pump using the...
-
The stockholders' equity of Zhou Corporation at January 1 follows: The following transactions, among others, occurred during the year: Jan. 1 Announced a 4-for-1 common stock split, reducing the par...
-
The following adjusted revenue and expense accounts appeared in the accounting records of Pashi, Inc., an accrual basis taxpayer, for the year ended December 31, Year 2. Revenues Net sales $3,000,000...
-
SD05 4) What application control and on what field lets you know the unit of currency of the amount
-
Why is a new infrastructure needed for GPRS, but not for HSCSD? Which components are new and what is their purpose?
-
For each of the following, identify the product (represented by A, B, C, and D) that would be formed through the indicated sequence of steps from the given starting material. O (1) mCPBA (2) EtMgBr...
-
Predict the major product from each of the following reactions. H O (1) (2) HO+ (3) HCrO4 MgBr (excess)
-
Below is a series of cost of goods sold sections for companies B, M, 0 , and S. Instruction Fill in the lettered blanks to complete the cost of goods sold sections. Beginning inventory Purchases...
-
Review and research this Guard RFID product technology. Real-Time Visibility of Your Staff, Patients, and Assets - GuardRFIDA Real-Time Location System (RTLS) is a precise positioning system that can...
-
How does your participation compare to your statements about collaboration measure up? Did the personal characteristics assist you with participating in class? How do your peers see your...
-
Fedex and UPS Documentary YouTubeLinks to an external site. Please Post Your Detailed Answers to the Following Questions: 1. What are the shipping performance measures of Fedex and UPS? 2. What...
-
Rocky Mountain Corporation makes two types of hiking boots-Xactive and Pathbreaker. Data concerning these two product line appear below: Direct materials per unit Direct labor cost per unit Direct...
-
1. Why was the project initiated? How does the project align with the mission and vision of the organization? What is the business case? 2. What is the scope of the project? What are the identified...
-
Given the information in Problem 10, what was the average real risk-free rate over this time period? What was the average real risk premium? In Problem 10 For Problem 9, suppose the average inflation...
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
What reagents and reaction conditions could be used to carry out the followingtransformations? Br - CH,CH,CH CH=CH, a) CH;CH,CH,CH;CH2 CH3 CH3 b) CH,CCI CH,COCH, CH3 CH3 C, Ph CH3 c) PHCH CI C=C CH3...
-
Suggest the best way to prepare these alkenes from alkylhalides. b) CH,CH CH,CH=CHCH_CH3 a) CH,CH,CH,CH,CH=CH, CH3 CH3 d)
-
When heated in ethanol, this alkyl halide gives two substitution and two elimination products. Show the structures of these products and the mechanism for theirformation. Br ELOH
-
The 6 - month LIBOR is 1 . 6 5 % . ( a ) calculate the 6 - month discount factor ( b ) calculate the 6 - month zero - coupon price of a $ 1 bond ( c ) calculate the effective annual yield for that...
-
4. Multi Step Binomial Tree: Consider again the European call option with three months left to maturity written on a non-dividend paying stock. As in exercise 2a, let today's stock price be 100 kr,...
-
C 0 Keenan Industries has a bond outstanding with 25 years to maturity, a 7.45% nominal coupon, semiannual payments, and a $1,000 par value. The bond has a 5.90% nominal yield to maturity, but it can...

Study smarter with the SolutionInn App


