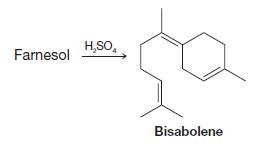
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for
Question:
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for this reaction.
Transcribed Image Text:
Farnesol H₂SO4 Bisabolene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Solution The proposed mechanism for farnesol to bisabolene ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
When 2-methyl-2-propanol is treated with sulfuric acid, 2-methylpropene is formed. Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of...
-
A possible mechanism for the pathology of trinucleotide repeat diseases is aberrant translation in which all three reading frames are used. Which repeating amino acid residues will result from the...
-
A possible mechanism for a gas-phase reaction is given below. What is the rate law predicted by this mechanism? (fast equilibrium) 2NOCI NOC NO2NOCI (slow)
-
Suppose that you are holding your toy submarine under the water. You release it and it begins to ascend. The graph models the depth of the submarine as a function of time. What is the domain and...
-
Repeat Exercise 29, but let n be the number of collisions (in thousands) for the year that is t years since 1985. Which of your responses for this exercise are the same as those for Exercise 29?...
-
Mishima, Inc. indicated in a recent annual report that approximately $19 million of merchandise was received on consignment. Should Mishima, Inc. report this amount on its balance sheet? Explain.
-
A distillation column with \(8.01 \mathrm{~m}\) of packing operating at total reflux separates methanol from ethanol at \(101.3 \mathrm{kPa}\). Average relative volatility is 1.69 . Methanol mole...
-
Daves Restaurant is a chain that employs independent evaluators to visit its restaurants as customers and assess the quality of the service by filling out a questionnaire. The company evaluates...
-
Escenario 1 : Los aos 2026 y 2027 el tipo de cambio se mantiene en 800 $/US$. Adems, los Gastos Administrativos crecen un 10 % durante el ao 2026 en relacin al ao 2025, y durante le ao 2027 crecen un...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
When morphine reacts with 2 mol of acetic anhydride, it is transformed into the highly addictive narcotic heroin. What is the structure of heroin?
-
One of the important steps in Gates synthesis of morphine involved the following transformation: Suggest how this step was accomplished. CHO CHO NC CHO CHO NC-
-
Describe the desirable qualities companies should consider when choosing product names.
-
Students will research the history and development of macOS, looking at the major differences between the macOS Versions. Choose at least three macOS versions to compare. Take versions from three...
-
What happens when someone does not comply with a departure order? You are inadmissible What is the removal order if someone is found inadmissible due to criminality? Does a younger offender have an...
-
Changes in Current Operating Assets and Liabilities Blue Circle Corporation's comparative balance sheet for current assets and liabilities was as follows: Dec. 31, Year 2 Dec. 31, Year 1 Accounts...
-
Wyler is preparing to conduct two field tests for individual semi-structured interviews with participants who meet the inclusion criteria for his study via video conferencing. How should Wyler...
-
Simplify. (3x x + 4) (2x +7x + 1) + (9x-3x+2) -
-
Sydney Retailing (buyer) and Troy Wholesalers (seller) enter into the following transactions. Both Sydney and Troy use a perpetual inventory system and the gross method. May 11 Sydney accepts...
-
Use multiplication or division of power series to find the first three nonzero terms in the Maclaurin series for each function. y = e x2 cos x
-
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain. , , . -C-C-C-
-
Construct a tree diagram for the absorption of Hm assume that Jam . . - -C-
-
Predict the multiplicities of the absorptions for the hydrogen's of these groups, assume that hydrogen's labeled a are different from those labeled x but that all of those labeled a are identical and...
-
2 . 5 . 1 . The ARS / EUR Bid is 9 1 8 . 8 5 0 0 and the Offer is 9 9 4 . 1 0 5 5 . ( Our EUR is the base currency in this notation ) . What will the mid rate then be for the EUR / ARS quotation? (...
-
Calculate the expected standard deviation on stock: State of the economy Probability of the states Percentage returns Economic recession 11% -4.2% Steady economic growth 35% 3.3% Boom Please...
-
You expect Sharp Steel Company to pay a dividend of $2.37 per share next year. You expect the dividend to grow 10% the following year, 7% the year after that, and then level off to a growth rate of...

Study smarter with the SolutionInn App


