Are the following pairs of structures resonance contributors or different compounds? a. b. CH 3 CH=CHCHCH=CH 2
Question:
Are the following pairs of structures resonance contributors or different compounds?
a.
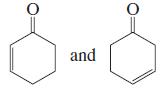
b. CH3CH=CHĊHCH=CH2 and CH3ĊHCH=CHCH=CH2
c.

d.
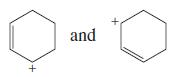
e. CH3C+HCH=CHCH3 and CH3CH=CHCH2C+H2
Transcribed Image Text:
and
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
8888 Different compounds a b HC CH CH CH CH CH2 and HC CH CH CH CH CH and d ...View the full answer

Answered By

Shyam Kumar
I have done B.Tech in chemical engineering .I have teaching high school level students and for engineering/medical entrance exams like IIT,NEET etc I have very good experience of dealing with all kinds of students weaker and brilliant and how to develop interest among students by representating the topics in unique way so that they can learn without stress and enjoy the subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the relationship between the following pairs of structures. The possible relationships are: same compound cis-trans isomers constitutional isomers (structural isomers) not isomers (different...
-
Give the relationships between the following pairs of structures. The possible relationships are: same compound, cis-trans isomers, constitutional (structural) isomers, not isomers (different...
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
How to respond to this response The effective utilization of data is integral to enhancing efficiency and making informed decisions?
-
Calculate the Doppler linewidth for the 589-nm line of Na and for the 254-nm line of Hg, both at 2 000 K.
-
The financial statements of MNE Corporation revealed the following: Required 1. Why does MNE Corporation use foreign exchange derivatives? What is the likely form of these derivatives? 2. What is the...
-
On January 1, 2020, lessor Marcy and lessee Lenox contract for the lease of a machine for five payments of \(\$ 7,000\) each. The \(\$ 7,000\) payments are to be paid at the end of each year. They...
-
Lowell Companys manufacturing overhead budget for the first quarter of 2012 contained the following data. Actual variable costs were: indirect materials $13,800, indirect labor $9,600, utilities...
-
Suppose you take out a 30-year mortgage for a house that costs $463,200. Assume the following: The annual interest rate on the mortgage is 4.6%. The bank requires a minimum down payment of 12% at...
-
WAR (We Are Rich) has been in business since 1987. WAR is an accrual-method sole proprietorship that deals in the manufacturing and wholesaling of various types of golf equipment. Hack & Hack CPAs...
-
Draw resonance contributors for the following ions: a. b. c. d.
-
The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated and stored. Why is this carbocation so stable? C. triphenylmethyl chloride
-
The mechanical tachometer measures the rotational speed N of the shaft by the horizontal motion of the collar B along the rotating shaft. This movement is caused by the centrifugal action of the two...
-
In a research report, a student is running a two-variable OLS regression to prove that Chinese firms' ROA (a traditional proxy for firm profitability) is adversely affected by CSR spending (measured...
-
Preble 1. (13 points) The ACCT 240 Co. had the following financial information for 2022: Average Cash-C Average Current Liabilities $1OK Average Accounts Receivable $10K, Average Inventory - SISK:...
-
Calculate Marginal cost of inventories. Alabang Company's records for the year ended December 31 showed that there were no work-in process inventories at the beginning and end of the year: Net Sales...
-
The order-to-delivery process for a manufacturing company, Company ABC, can be summarized as follows A quality issue faced by this company is late deliveries to customers. (a) Select any three (3)...
-
Consider 40,000 Ib/h (18,150 kg/h) of a 25 wt.% MgSO4 solution being fed at 200F (366K) to an evaporative crystallizer that boils off water at a rate of 15,000 Ib/h (6800 kg/h). The crystallizer is...
-
Your friend is just finishing a study of attendance at Los Angeles Laker regular-season home basketball games when she hears that youve read a chapter on serial correlation and asks your advice....
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Give the m/z values of the fragment ions expected from b - type fragmentation of an M + 1 ion of the peptide N - F - E - S - G - K
-
Give the curved-arrow mechanism for the formation of each of the following fragment ions in Fig. 26.5 from an M + I ion. The fragment at m/z = 551.94
-
Using the curved-arrow notation, write in detail the mechanisms for the reactions in (a)Eq. 26.41a
-
i don't understand this Sample Java output: (printList = "y") Please enter the list length: 7 Key = 3 Unsorted list: 5 4 2 3 3 1 4 Key 3 found in linear search at position: 4 Sort time is 0 mi...
-
Sheridan dish printery publishes the best-selling captain Cajun cookbook that sells for $9. The company incurs variable cost a $4 per cookbook and total fixed cost of $250,000. If the companies tax...
-
I do not know what else do you need I only need help to solve this because I do not know how to. The question is not incomplete and I already gave your all the relevant information. This i...

Study smarter with the SolutionInn App


