Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? (a)
Question:
Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides?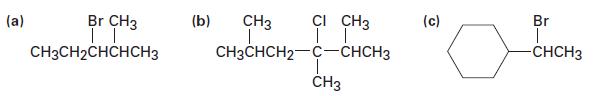
Transcribed Image Text:
(a) Br CH3 | | CH3CH2CHCHCH3 (b) CH3 CI CH3 | | CH3CHCH2-C-CHCH3 CH3 (c) Br -CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To determine the products of elimination reactions of the given alkyl halides we need to identify the type of elimination reaction that will take plac...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ignoring double-bond stereo chemistry, what products would you expect from elimination reactions of the following alkyl halides? Which will be the major product in eachcase? Br CH CH CI CH (a) (b) Br...
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
In Exercises 7-9, find the indicated measure. The area of a circle is 380 square inches. Find the radius.
-
You are the manager of a large but privately held online retailer that currently uses 17 unskilled workers and 6 semiskilled workers at its warehouse to box and ship the products it sells online....
-
Jack Backpack Company manufactures a variety of backpack models. Each backpack requires a variety of direct materials, including fabric, buckles, zippers, and other materials. Company managers have...
-
Pablo S.A. is a manufacturer of children's shoes, located in Spain. It owns 51 percent of the voting stock of Stabifoot, a specialty shoe manufacturer located in Belgium. Pablo acquired its interest...
-
Austen Ren owns a small retail ice cream parlor. He is considering expanding the business and has identified two attractive alternatives. One involves purchasing a machine that would enable Mr. Ren...
-
please organize carefully In the table below you will find simplified consolidated balance sheets for the chartered banking system and the Bank of Canada. Use columns 1.3 to indicate how the balance...
-
Name the following alkyl halides: (a) H3C Br Br | | || CH3CHCHCHCHCHCH3 CH3 (d) CHBr CH3CHCHCHCHCH3 (b) I CH3CH=CHCH2CHCH3 (e) CICHCHCHC=CCHBr (c) Br CI CH3 | || CH3CCHCHCHCH3 CH3
-
Assign configuration to the following substrate, and show the stereochemistry and identity of the product you would obtain by S N 1 reaction with H 2 O (reddish brown = Br):
-
Company A1 intends to raise $3 million by either of two financing plans: Plan A: Sell 100,000 shares of stock at $30 net to firm Plan B: Issue $3 million in long term bonds with a 10 percent coupon...
-
Revived Routines: Consumers are settling into new schedules and navigating a return to reality. They are eager to get on with their lives despite the uncertainties ahead. Using your knowledge and...
-
2. Suppose a business takes out a $300,000, ten years loan at 8 percent. The business wants to make equal principal payments over the period. Prepare an amortization schedule for this loan.
-
A patient present with dehydration, hypotension and fever. Lab testing reveals hyponatremia, hyperkalemia and hypoglycemia. These imbalance are corrected, burt the patient returnes 6 weeks later with...
-
At the end of this month, Leslie will start saving $200 a month for retirement through his company's superannuation plan. His employer will contribute an additional $0.50 for every $1.00 that he...
-
What is the bond's Macaulay duration of a2.5 years, 8 percent semiannual coupon bond selling at par of $1,000?
-
How do tariffs and quotas differ from one another? Identify the different forms each can take.
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
Consider the following reaction: a) How would the rate be affected if the concentration of tertbutyl bromide is doubled? b) How would the rate be affected if the concentration of sodium ethoxide is...
-
Beginning with Equation (5.5), use Equation (5.6) to eliminate V c and V d to arrive at the result w cycle = nR(T hot T cold )lnV b /V a .
-
Consider the following reaction: a) How would the rate be affected if the concentration of tertbutyl bromide is doubled? b) How would the rate be affected if the concentration of ethanol is doubled?...
-
TRUE OR FALSE: "Built" costumes are created in a costume shop, where "pulled" costumes are previously existing outfits selected by a costume designer Group of answer choices True False
-
Accounting allows businesses to communicate information about a company's financial performance to the following groups of users: Question 15Answer a. internal users, external users, and the...
-
Because the Medical Unit may perform some of the same tasks, close coordination between the Medical Unit and the Compensation/Claims Unit is essential. The Compensation/Claims Unit maintains logs on...

Study smarter with the SolutionInn App


