Predict the products you would expect from the following reactions. Indicate the major product in each case.
Question:
Predict the products you would expect from the following reactions. Indicate the major product in each case.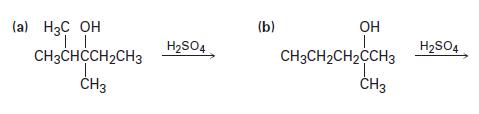
Transcribed Image Text:
(a) H3C OH TL CH3CHCCH₂CH3 T CH3 H₂SO4 (b) OH I CH3CH₂CH₂CCH3 CH3 H₂SO4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Lets predict the major products for each reaction Reaction 1 HBr red Br In this reaction HBr adds ac...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products you would expect from the reaction of NaBH4 with the following compounds. You may assume that these reactions take place in methanol as the solvent. (a) CH3--(CH2)8--CHO (c) Ph...
-
Predict the products you expect when the following starting material undergoes oxidation with an excess of each of the reagents shown below. (a) Chromic acid (b) PCC (Pyridinium Chlorochromate) (c)...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
In Problems 67 90, multiply the polynomials using the special product formulas. Express your answer as a single polynomial in standard form. (x-4)
-
Recently, the owner of a Trader Joes franchise decided to change how she compensated her top manager. Last year, she paid him a fixed salary of $ 65,000, and her store made $ 120,000 in profits (not...
-
The luxury-goods manufacturer Louis Vuitton has several factories in the United States, France, and Spain that produce its bags and accessories. Part of the Louis Vuitton brand appeal to consumers is...
-
The impartial game Cram is played on a board of \(m \times n\) squares, where players alternately place a domino on the board which covers two adjacent squares that are free (not yet occupied by a...
-
Mendosa Company closes its books monthly. On September 30, selected ledger account balances are: Notes Receivable.. $33,000 Interest Receivable... 170 Notes Receivable include the following. Interest...
-
You will be employed in the finance department of Tracy Show Hire Pty Ltd in the Victoria Headquarter, and you need to complete the lodgment schedule below to ensure you meet all the businesses...
-
How might you prepare the following substances by using nucleophilic substitution reactions? (a) CH 3 CH 2 CH 2 CH 2 OH (b) (CH 3 ) 2 CHCH 2 CH 2 N 3
-
Show the products obtained from addition of CH 3 MgBr to the following carbonyl compounds: (a) (b) (c) || CH3CHCHCCHCH3
-
Research the AICPA's Code of Professional Conduct (which is available on the AICPA's website - www.aicpa.org) to research what it means to be in a "network" of firms. How might the actions of one of...
-
In December, T.D. Weber evaluated his stock portfolio and found that his DLW Inc. common stock had declined in value since he purchased it. He decides to sell the DLW stock on December 15. However,...
-
Flowers Ltd. prepares and sells large flower bouquets for all occasions. Currently, workers at the production shop produce baskets that are used in each flower arrangements. Based on annual sales of...
-
Performance Running Company is a company specialized in manufacturing running equipment. You are working as a marketing officer with Performance Running Company. You like your job and you adore your...
-
Along with burritos, Chipotle has cooked up profitable growth for many years. The company's build-your-own meal model and focus on organic and naturally raised ingredients successfully attracted...
-
CaniRug will begin to produce and sell one-size-fits-all wigs in two colors: blonde and brunette. The brown wig is projected to be the better seller with sales of 100 wigs per week, whereas the...
-
Discuss which industries are Canadas main export industries based on national competitive advantage.
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
When 2-chloro-1, 1, 2, 3, 3-pentamethylcyclohexane is treated with sodium hydroxide, neither E2 nor S N 2 products are formed. Explain.
-
Why are the triple point temperature and the normal freezing point very close in temperature for most substances?
-
Identify the major and minor product(s) that are expected for each of the following reactions: a. b. c. d. e. f. g. h. i. j. k. l. m. n. OTs Naci DMSO NaOH
-
The pension plan assets balance is reported Multiple choice question. in the disclosure notes. in the income statement. in the balance sheet. in the statement of cash flows
-
Entering notes is the main function of notation apps. Higher price solutions like Finale and Sibelius offer a lot of entry options. Which item(s) below are not included? Group of answer choices...
-
Companies with relatively high assets-to-sales ratios (= A*/S) require a relatively large amount of new assets for any given increase in sales; but that doesn't always imply that they have a greater...

Study smarter with the SolutionInn App


