What alcohols might the following alkenes be made from? (a) CH3 CH3 (b) CH3CHCH=CHCHCHCH3
Question:
What alcohols might the following alkenes be made from?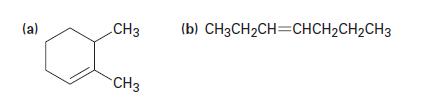
Transcribed Image Text:
(a) CH3 CH3 (b) CH3CH₂CH=CHCH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine the alcohols that the given alkenes might be made from we need to identify the possible ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b) CHH (c) .CH- CHCHCH2CH2OH
-
What alkenes might the following alcohols have been prepared from? (b) (a) CH3CCH2CH2CH2CH3 C
-
What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2 "CH
-
In Problems 25 54, solve each system. Use any method you wish. 2xxy + y = 8 xy = 4
-
A firm has $ 1.5 million in sales, a Lerner index of 0.57, and a marginal cost of $ 50, and competes against 800 other firms in its relevant market. a. What price does this firm charge its customers?...
-
Dominion Industries manufactures three models of a product in a single plant with two departments: Cutting and Assembly. The company has estimated costs for each of the three product models: the...
-
Consider the game Cram from Exercise 1.4, played on a \(1 \times n\) board for \(n \geq 2\). Let \(D_{n}\) be the Nim value of that game, so that the starting position of the \(1 \times n\) board is...
-
Using the data in the student spreadsheet file P&G.xlsx (to find the student spreadsheets for Financial Analysis with Microsoft Excel, seventh edition, go to www.cengagebrain.com) forecast the June...
-
Firm A is a publicly traded company. It has 7.1 million sharesoutstanding and the current price of each share is $15.0. Firm Aalso has 100,000 annual coupon bonds. Half of these bonds have 5years t 2...
-
What effect would the following changes have on the rate of the S N 1 reaction of tert-butyl alcohol with HBr? (a) The HBr concentration is tripled. (b) The HBr concentration is halved, and the...
-
What product would you expect to obtain from the S N 2 reaction of (S)-2 bromohexane with sodium acetate, CH 3 CO 2 Na? Show the stereochemistry of both product and reactant.
-
Find in the network shown using PSPICE. 12V
-
If a business combination occurs and the consideration paid exceeds the fair value of the identifiable net assets of the subsidiary on the acquisition date and the parent acquires less than 100% of...
-
What is the management plan in a patient with secondary polycythaemia presenting with transient ischaemic attacks (TIA)? Question 33 Why do patients with polycythaemia vera have a tendency to bleed,...
-
The Personnel Department at Hernandez Bros. is centralized and provides services to the two operating units: Miami and New York. The Miami unit is the original unit of the company and is well...
-
Muntjac Corporation's WACC is 14% and its tax rate is 40%. Muntjac's pre-tax cost of debt is 15% and it's debt-equity ratio is 1:2. If the risk-free rate is 9% and the market rate of return is 12%,...
-
14. What is meant by maturity intermediation? (LG 1-6)
-
Explain the need for customer knowledge, following clients, and following rivals in the FDI decision.
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
Identify whether each of the following reagents would be a strong nucleophile or a weak nucleophile, and also indicate whether it would be a strong base or a weak base: a.
-
We have seen that NaH is a strong base but a weak nucleophile. In contrast, lithium aluminum hydride (LAH) is a reagent that can serve as a source of nucleophilic hydride ion: In this case, LAH...
-
a) NaOH is a strong nucleophile and strong base. The substrate in this case is primary. Therefore, we expect S N 2 (giving the major product) and E2 (giving the minor product). b) NaSH is a strong...
-
The status of _______________ organizations are held in trust, rather than owned; there are many hospitals that fit into this tax status. Group of answer choices for-profit not-for-profit All of...
-
hich of the following is not considered an equity-based compensation for executive management? a. Stock options b. Restricted stock units (RSUs) c. Restricted stock awards d. Deferred compensation...
-
The objective of effective financial reporting is to provide information in a(n) ____________ manner that will assist in making decisions regarding the allocation of resources for the organization....

Study smarter with the SolutionInn App


