Assuming the D configuration, identify A and B. an aldopentose A Kiliani-Fischer synthesis an aldohexose B (one
Question:
Assuming the D configuration, identify A and B.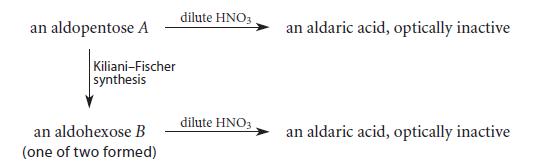
Transcribed Image Text:
an aldopentose A Kiliani-Fischer synthesis an aldohexose B (one of two formed) dilute HNO3 dilute HNO3 an aldaric acid, optically inactive an aldaric acid, optically inactive
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The aldopentose A is Dribose and the aldohexose B is Dallose T...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Plymouth Company holds a 90% interest in Savannah, Inc., which was acquired in a previous year. As of the end of the current fiscal period, the following information is available: Savannah Plymouth...
-
a State the electronic configurations of the following atoms or ions: i. Ti ii. Cr iii. Co iv. Fe 3+ v. Ni 2+ vi. Cu + b. Explain why scandium (which forms only one ion, Sc 3+ ) and zinc (which forms...
-
Use data from Appendix D to determine (a) r H, r S, and r G at 298 K and (b) K at 875 K for the water gas shift reaction, used commercially to produce H 2 (g): CO(g) + H 2 O(g) CO 2 (g) + H 2...
-
Implement a Digraph data type that represents directed graphs, where the direction of edges is significant: \(\operatorname{addEdge}(v, w)\) means to add an edge from \(v\) to \(w\) but not from...
-
Show that if a particle moves at an angle with the x axis with speed ?u in frame S, it moves at an angle with the x axis in S given by tan 0' r(cos 0 - Vlu) sin 0
-
Solve the proceeding problem for an aluminum plate with b = 12 in., t = 1.0 in., E = 10,00 ksi, v = 0.33, Px = 90 k, Py = 20 k, and V = 15 k?
-
Why is a yield curve showing higher long-term interest rates than short-term rates (rising curve) called a normal curve?
-
CH3CH2SH + CH3O- A (contains sulfur) + B C + H2O D + E (which is inorganic) (a) Given the above sequence of reactions, draw structures for A through E. (b) Rewrite the reaction sequence, showing all...
-
A manufacturer incurs the following costs in producing x water ski vests in one day, for 0
-
Draw a Fischer projection for the aldaric acid, and a structure of the 1,4-lactone, derived from the oxidation of (a) D -galactose; (b) D -mannose.
-
Give Fischer projections for the aldaric acids derived from both D-glucose and L- gulose. What is the relationship between these structures?
-
Return on investment may be separated into two components. Name them and describe what each can tell you.
-
Consider the challenging conditions that you would face in the situation presented. What would your next step be in confronting the HR challenge faced by the hospital from a competitive standpoint?...
-
A SOP data given in the Excel file. By reading following information, answer appropriately to the questions a) For the data that you have, provide both level and chase. Discuss the main...
-
What mass (in grams) of sodium hydroxide (NaOH, molar mass = 40.0 g.mol-1) is needed to make 57.6 mL of a 0.122 M NaOH solution?
-
Using the scenario provided, respond to the critical-thinking questions. Scenario A nurse employed by a temporary agency is assigned to a neurology unit for a 12-hour shift. On arrival, she discovers...
-
Nykaa is an Indian e-commerce platform that specializes in beauty and wellness products. Founded in 2012 by Falguni Nayar, a former investment banker, the company has become one of the largest...
-
From chapter 14, figure 14.2 a. How does this aspect of the bodys defenses help protect against Streptococcus pneumoniae infection? Nasal cavity Nostril Oral cavity Pharynx Epiglottis Larynx Trachea...
-
Write each fraction as a percent. 7 50
-
Which of the following statements would correctly de-scribe the isoelectric point of cysteic acid, an oxidation product of cysteine? Explain your answer. (1) Lower than that of aspartic acid (2)...
-
Dansyl chloride (5-dimethylamino-1 -naphthalenesul- fonyl chloride) reacts with amino groups to give a fluorescent derivative. After a peptide P with the composition (Arg,Asp,Gly,Leu2,Thr,Val) reacts...
-
The peptide hormone glucagon has the following amino acid sequence: His-Ser-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Ser-Arg-Arg-Ala-Gln-Asp- Phe-Val-Gln-Trp-Leu-Met-Asn-Thr Give the...
-
Louis files as a single taxpayer. In April of this year he received a $910 refund of state income taxes that he paid last year. How much of the refund, if any, must Louis include in gross income...
-
6 Lester and Forrest Scruggs have presented you with the following information for Scruggs, Inc. For 2021 they would like for you to 7 complete a(n) 1 adjusted Trial Balance (prepare this on a...
-
A small departmental store buys and sells products from various companies. They buy some of the products at discounted prices when they order large numbers. They sell all their products 20% above the...

Study smarter with the SolutionInn App


