Consider the following mechanism for Eq. 18.19. Identify the process associated with each step. Counting electrons at
Question:
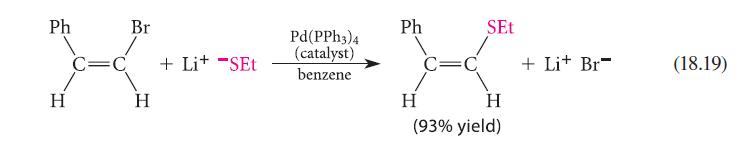
Consider the following mechanism for Eq. 18.19. Identify the process associated with each step. Counting electrons at each stage may help you.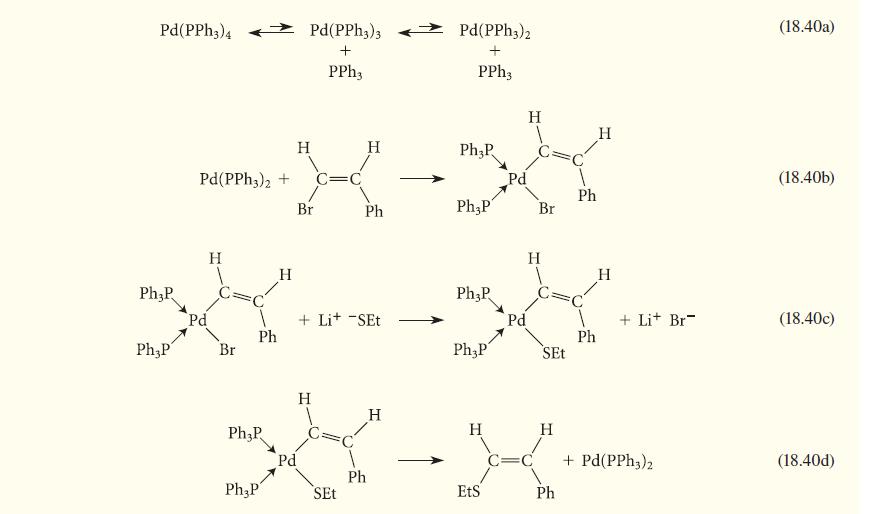
Transcribed Image Text:
Pd(PPH3)4 Ph3P Ph,P Pd(PPh 3)2 + Pd H Br Ph H Ph₂P Pd(PPH3)3 + PPh3 H Br C=C H Ph + Lit -SET Pd(PPH3)2 + PPh3 Ph₂P Ph,P Ph₂P Ph P Pd Pd H Br H SEt XXX-X Ph Ph H H + Lit Br + Pd(PPH3)2 (18.40a) (18.40b) (18.40c) (18.40d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Step 1840a consists of two successive ligand dissociations that reduce the ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Dickens, Kristen, is enrolled as a doctoral student in the Counselor Education at the University of New Orleans. She is a registered counselor intern in the state of Louisiana and works at a...
-
Implement the method contains() for HashST.
-
1. For the principal quantum number n = 3, what are the possible values of the quantum numbers and m? 2. What is the energy of the shortest wavelength photon emitted by the hydrogen atom?
-
The figure shows an idealized structure consisting of rigid bars ABC and DEF joined by linearly elastic spring ( between C and D. The structure is also supported by translational elastic support ( at...
-
What does TSR correspond to in terms of investment choice?
-
Urban Auto Glass specializes in the repair and replacement of windshields for passenger vehicles. Variable and fixed costs related to installation activities for the most recent month (July) are...
-
-10 -8 -6 T -20 2 -2 2 4 6 8 10 AA -4 6 -10 h(t) -12 -14 Find a trigonometric function (sin or cos) for the function shown above.
-
Arrange the following compounds according to increasing rate of elimination with NaOEt in EtOH. What is the product in each case? Ph H C=C A Ph Br Ph H C=C B Br Ph Ph H C=C C Ph Cl H H =C D Br Ph
-
The reaction given in Fig. P17.59 occurs by a mechanism called the S N 2 mechanism, which is a bimolecular substitution that occurs by reaction of the nucleophile at an allylic carbon. In this...
-
A firm reported the following income statement (amounts in millions): Net sales ......................................................... $496 Cost of goods sold...
-
A shoe store's records show that 30% of customers making a purchase use credit cards to make payment. This morning, 20 customer purchased shoes from the store. a) What is the probability that at...
-
Use the Android Studio terminal window to log into the phone using "adb shell". From the command line, type "run-as package-name". Then navigate to "/data/data/package-name/shared_prefs", and display...
-
Single and age 5 3 is a participant of his employer's qualified profit - sharing plan I would like to make an IRA contribution If AGI is 8 1 , 0 0 0 ( all comprised of W - 2 earnings and portfolio...
-
CGWC is being sued by a former employee for wrongful dismissal. The employee is suing CGWC for $40,000, and the case is currently in mediation. Legal counsel suggests that it is unlikely that $40,000...
-
Health Cooperative is an outpatient surgical clinic. It budgets $691,900 of overhead cost for the year. The two main surgical units and their data follow. Service General surgery Orthopedic surgery...
-
From chapter 9, figure 9.24 d. Use the technology from figure 10.11 as your guide to arrive at the same result as shown here, but without the mouse. Figure 10.11 Live R strain Heat-killed S strain...
-
During registration at Tech every quarter, students in the Department of Management must have their courses approved by the departmental advisor. It takes the advisor an average of 4 minutes...
-
A chemist finds that the addition of (+)-epinephrine to the catalytic reduction of butan-2-one (Figure 5-16) gives a product that is slightly optically active, with a specific rotation of +0.45o...
-
1. Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. (a) 1-bromo-1-chloroethane (b) 1-bromo-2-chloroethane...
-
Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through...
-
The European Commission rebuked Greece in 2010 for falsifying public finance data. Which of the following statements is the reason for inaccurate information in the above situation? Group of answer...
-
Discounted Cash flow dan Non Discounted Cash flow. Jelaskan apa maksud dan perbedaan dari dua konsep tersebut. Metode kriteria analisis investasi apa saja yang termasuk dalam masing-masing konsep...
-
Bowling Green Financial is preparing its Budgeted Income Statement for next year. Bowling Green Financial has collected the following information about this statement: Sales for next year total...

Study smarter with the SolutionInn App


