In an effort to duplicate in the laboratory the natural processes that resemble the cyclization of 2,3-oxidosqualene,
Question:
In an effort to duplicate in the laboratory the natural processes that resemble the cyclization of 2,3-oxidosqualene, chemists in 1970 and 1980 carried out the conversions shown in Fig. P17.51. Give a curved-arrow mechanism for each. (For purposes of this problem, let the acid and base catalysts be a general acid A—H and its conjugate base A¯.)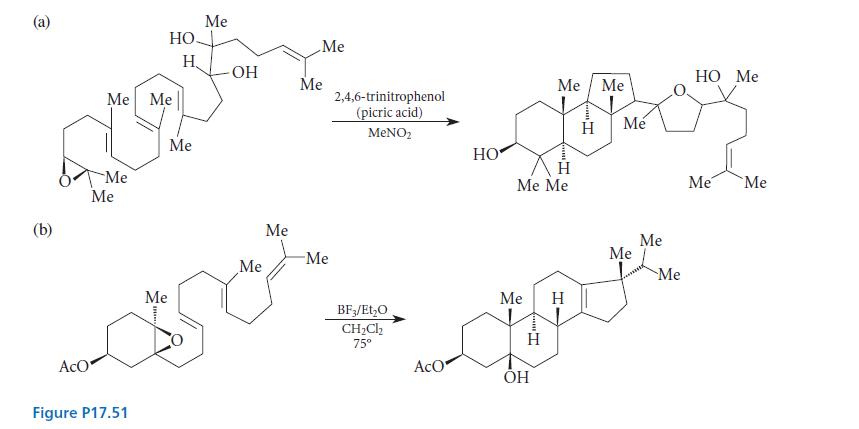
Transcribed Image Text:
(a) (b) Me Me AcO Me HO. Me DURA H Figure P17.51 Me Me -OH Me Me Me Me anor Me -Me 2,4,6-trinitrophenol (picric acid) MeNO BF3/EtO CHCl 75 HO AcO Me Me 1" Me Me OH " H M Me Me Me H Opp Me HO Me Me Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a HO Me Me Me Me Me Me Me A Me ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Chief Justice Truepenny (Guilty): Our murder statute permits no exception applicable to this case, however our sympathies may incline us to make allowance for the tragic situation in which these men...
-
Harley-Davidson: Preparing for the Next Century There are very few products that are so exciting that people will tattoo your logo on their body. Richard Teerlink, Retired CEO, Harley-Davidson In...
-
Two popular methods of financial statement analysis are horizontal analysis and vertical analysis. Explain the difference between these two methods. Discuss.
-
The compressor of a large gas turbine receives air from the ambient at 95 kPa, 20C, with a low velocity. At the compressor discharge, air exits at 1.52 MPa, 430C, with velocity of 90...
-
Marketing communicators aim to build customer-brand relationships and guide customers through the five As of the customer journey. What is the first of the five As?
-
Inland Corporation issued 30,000 shares of \(\$ 5\) par value common stock at \(\$ 15\) per share and 8,000 shares of \(\$ 50\) par value, eight percent preferred stock at \(\$ 85\) per share. Later,...
-
The following information concerns the intangible assets of Epstein Corporation: a. On June 30, 2011, Epstein completed the purchase of the Johnstone Corporation for $2,000,000 in cash. The fair...
-
Metlock Merchandising uses an aging schedule to determine its estimated uncollectible accounts at year end. The percentage estimates of bad debts are as follows: METLOCK MERCHANDISING Aged Schedule...
-
One interesting use of DielsAlder reactions is to trap very reactive alkenes that cannot be isolated and studied directly. One compound used as a diene for this purpose is diphenylisobenzofuran,...
-
Calculate n in the d n notation for ferrocene, Cp 2 Fe.
-
Complete the payroll register of T Rocks showing the total gross for the week ended 19 February 2022. Use the information shown in figure 14.4. Payroll Summary of T Rocks for week ended 19 February...
-
Of the sundaes recently sold at Ice Cream Haven, 4 had nuts and 20 did not. Considering this data, how many of the next 84 sundaes sold would you expect to have nuts?
-
A thin plate with the shape shown in figure below is subjected to 10 kN load. The modulus of elasticity of the material is 100 GPa. The plate thickness is 2.mm. Develop the element-nodes table. Write...
-
Tina can mow a lawn by herself in 4 hours. Bill can mow the same lawn by himself in 5 hours. How long would it take the to mow the lawn if they work together?
-
Key West, Florida to Seattle, Washington is 3,518 miles. If it takes 7 hours and 15 minutes to fly there, what is the average speed?
-
Define and explain probability terminology, likelihood and experiments Question If a six-sided die is rolled one time, and A is the event of rolling a number less than 7 . Find the probability of...
-
A regional airline and a furniture manufacturer each generate annual revenue of $120 million and earn net income of $10 million. Which company probably has the higher breakeven point? Explain.
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
Show how you might use SN2 reactions to convert 1-chlorobutane into the following compounds. (a) Butan-1-ol (b) 1-fluorobutane (c) 1-iodobutane (d) CH3-(CH2)3-CN (e) (f) CH3CH2-O-(CH2)3-CH3 (g) CH3-...
-
For each pair, predict the stronger nucleophile in the SN2 reaction (using an alcohol as the solvent). Explain your prediction. (a) CH3CH2)3N or (CH3CH2)2NH (b) (CH3)2O or (CH3)2S (c) NH3 or PH3 (d)...
-
When diethyl ether (CH3CH2OCH2CH3) is treated with concentrated HBr, the initial products are CH3CH2Br and CH3CH2OH. Propose a mechanism to account for this reaction.
-
Marcus Corporation is currently all equity financed and has a value of $75 million. Investors currently require a return of 11.70 percent on common stock. Marcus pays no taxes. Marcus plans to issue...
-
(a) Why are there surprise market crashes? The market should signal this or at least communicate in a timely fashion. It could be said that insufficient attention has been paid to psychological...
-
Included in cost of sales is a reserve for a possible decline in the market value of finished goods inventory of $50,000. There is also an obsolescence reserve of $70,000 for some raw materials that...

Study smarter with the SolutionInn App


