Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20
Question:
Draw a plausible mechanism for each of the following transformations:
(a)
![[TSOH] MENH2 -H20](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1606/7/3/1/2195fc4c5d334dde1606731218633.jpg)
(b)
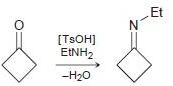
Transcribed Image Text:
[TSOH] MENH2 -H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (20 reviews)
For each of the mechanisms shown below the first two steps can be reversed first the am...View the full answer

Answered By

John Aketch
I am a dedicated person with high degree of professionalism, particularly in academic writing. My desire is to is to make students excel in their academic endeavor.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H,SO4] -H20 [H,SO4] -H20
-
Draw a plausible mechanism for each of the following transformations: a. b. c. d. e. Pyridine CI
-
Propose a plausible mechanism for each of the following reactions: a. b. stitl. [H,SO,] Conc. H2SO4
-
Lombard Ltd has been offered a contract for which there is available production capacity. The contract is for 20,000 identical items, manufactured by an intricate assembly operation, to be produced...
-
When writing technical reports, be aware that the numbers, formulas, and concepts can sometimes be overwhelming for the reader. Illustrate how to meet that challenge by doing the following: Identify...
-
Develop Eq. (12.35) for Sid by appropriate application of Eqs. (5.41) and (5.42) to a mixing process.
-
Describe two different methods used for calculating the value of a subscription right.
-
On December 31, 2017, before the books were closed, the management and accountants of Madrasa Inc. made the following determinations about three pieces of equipment. 1. Equipment A was purchased...
-
4. A plant asset acquired on October 1, 2021, at a cost of $6,000,000 has an estimated useful life of 10 years. The residual value is estimated to be $600,000 at the end of the asset's useful life....
-
Your company is planning to borrow $1 million on a 5-year, 15%, annual payment, fully amortized term loan. What fraction of the payment made at the end of the second year will represent repayment of...
-
Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20 Et [TSOH] EENH2 -H20
-
Using what you know about the expanded source table, fill in the missing values in the table shown here: df Source SS MS Gender 248.25 Parenting style 84.34 33.60 Gender X style Within 1107.2 36...
-
For a mortgage pass-through security, which of the following risks most likely increases as interest rates decline? A. Balloon B. Extension C. Contraction
-
What factors contribute to the formation of high-performance teams, and how can organizations cultivate and sustain these dynamics over time ?
-
Discuss factors that influence the level of cohesiveness within a group. (10 marks ) Critically examine the consequences of group cohesiveness to individuals and organization. (10 marks ) Political...
-
There are many social determinants factors that affect health status , discuss in your own opinions How does a systems approach could and should be utilized by healthcare organizations in trying to...
-
How do you define operational excellence? What factors are involved in achieving operational excellence? Who (within an organization) is responsible for operational excellence and why is this...
-
2) f(x) = x-3 g(x)=2x-1 (fg)(x): f(g(x)): 3) f(x)=2x+5 g(x) = 10(1.05)* (g-f)(2): g(f(x)):
-
In Figure 3.2, why do the intervals span from 0.5 to 1.5, from 1.5 to 2.5, etc., when all of the numbers in the dataset are whole numbers? Figure 3.2 1.5 2.5 5- Height of bar indicates 4 number of...
-
Pappa's Appliances uses the periodic inventory system. Details regarding the inventory of appliances at January 1, purchases invoices during the year, and the inventory count at December 31 are...
-
Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and so forth. (a) (b) CH3-C¡CH (c) (d) (e) (f) Obtained from oil of cloves -2. Sex attractant...
-
Boron trifluoride (BF3) has no dipole moment ( = 0 D). Explain how this observation confirms the geometry of BF3 predicted by VSEPR theory.
-
Identify all of the functional groups in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) Vitamin D3 HO OMe Aspartame O NH2 NH2 Amphetamine Me Cholesterol HO OCH2CH3 Demerol CH A...
-
Required information [The following information applies to the questions displayed below] Project Y requires a $336,000 investment for new machinery with a six-year life and no salvage value. The...
-
Exercise 17-5 (Algo) Product mix and plantwide rate versus activity-based costing LO P1, P3 Wess Company has limited capacity and can produce either its standard product or its deluxe product....
-
Check m Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year....

Study smarter with the SolutionInn App


