Draw all significant resonance structures for each of the following compounds: a. b. c. N' z-
Question:
a.
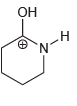
b.
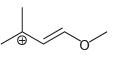
c.
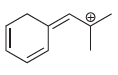
Transcribed Image Text:
Он н N' z-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all significant resonance structures for each of the following compounds: Testosterone Estradiol (Female sex hormone) (Male sex hormone)
-
Draw contributing resonance structures for each of the following species, and rank the structures in order of decreasing contribution to the hybrid: a. b. c. d. e. f. CH3C-CH CHCH3 CH3 0 CH3COCH3 +OH...
-
Draw resonance structures for each of the following radicals: (a) (b) (c) (d)
-
In Exercises evaluate the second derivative of the function at the given point. Use a computer algebra system to verify your result. (x) = cos x, (0, 1)
-
On April 31, 2016, Elkhorn Associates borrowed $10 million cash from Colonial Bank and issued a 5-month, noninterest-bearing note, priced to yield an effective interest rate of 10%. The stated...
-
If (1, (2, ( ( ( ( (n are the eigenvalues of A, find the eigenvalues and eigenvectors of A2.
-
What is meant by a value-added statement? To what extent is it likely to provide information over and above that already available in the income statement?
-
Hixson paid Galyen Petroleum Co. money he owed by issuing three checks to Galyen. The bank refused to cash the three checks because of insufficient funds in the Hixson account to pay all three....
-
5) Let f(x)=-2 In(x-5). a) Apply inverse functions to solve for the input(s) x that results in the output f(x) = -4.
-
Annie Campbell is a nurse on the evening shift from 10:00 P.M. to 6:00 A.M. at Community Hospital. She is responsible for 15 patients in her area. She averages two calls from each of her patients...
-
Each compound below exhibits one lone pair. In each case, identify the type of atomic orbital in which the lone pair is contained. a. b. c. .N.
-
Write a condensed structural formula for each of the following compounds: a. b. c. HO
-
Review the information in Illustration Capsule 4.1 concerning the value chain average costs of producing and selling an upscale polo shirt and compare this with the representative value chain...
-
(a) Write short notes on the following types of monetary standard that exist in developing countries: 1) Parallel standard(2 marks) ii) Bimetallic standard (2 marks) iii) Monometallic standard...
-
Create a C doe with a function that reads a single lowercase character, converts it to uppercase using a programmer-defined function, and then displays the uppercase equivalent character. Hint: Refer...
-
ABC Company purchased XYZ Company for $100 million in 2020.The fair value of XYZ's identifiable assets and liabilities at the acquisition date was $80 million.ABC Company has been amortizing goodwill...
-
Discuss some of the external factors that might affect a person's ability to detect a signal and how these external factors would affect detection rates. Use the terms hits, misses, false alarms, and...
-
A communication channel accepts X that is either 00 or 01 or 10 or 11. The channel transmits each bit binary input with probability p and enormously with probability (1-p) and gives out Y as output....
-
Give a pseudocode description of an algorithm for constructing a range tree from a set of n points in the plane in O(n log n) time.
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
In the mass spectrum of 2, 6-dimethyl-4-heptanol there are prominent peaks at m/z 87, 111, and 126. Propose reasonable structures for these fragment ions.
-
In the mass spectrum of 4-methyl-2-pentanone a McLafferty rearrangement and two other major fragmentation pathways occur. Propose reasonable structures for these fragment ions and specify the m/z...
-
What are the masses and structures of the ions produced in the following cleavage pathways? (a) A-cleavage of 2-methyl-3-hexanone (two pathways) (b) Dehydration of cyclopentanol (c) McLafferty...
-
Delta Company produces a single product. The cost of producing and selling a single unit of this product at the company's normal activity level of 108,000 units per year is: Direct materials Direct...
-
Explain to Khalid 1. When consolidation entry "G is prepared? 2. Give two reasons why is consolidation entry "G prepared? Assume an upstream intra-entity transfer. Khalid did not give an answer.
-
! Required information [The following information applies to the questions displayed below.] The following events apply to Gulf Seafood for the Year 1 fiscal year: 1. The company started when it...

Study smarter with the SolutionInn App


