Draw the products of each of the following acid-base reactions, and then predict the position of equilibrium
Question:
(a)
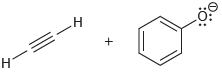
(b)
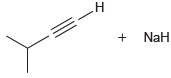
Transcribed Image Text:
н н NaH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
412+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) (e) Nal (1 mol) H -acetone GH,H + NaBr Nal (1 mol), CH,NaCl CI CI BrBr (1 mol C4HS2 2 NaBr OH NaH (-H2) Cl Et O...
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) Product of (c) heat (e) (f) (g) Aniline + propanoyl chloride (h) Tetraethylammonium hydroxide heat (i) p-Toluidine...
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. HCI CH CH2CH CHCH2OH excess catalytic CCH CH3 NH2NH2 CH2CH3 NH2NH 1. NaBH4 0 HCI excess CH,CH,CH,COCH CHs 2. H3o 1. LIAIH...
-
Discuss Planning & Preparation in relation to: Preparing work documents and performing document review Preparing an audit plan
-
The following information was gathered for Rogers Company for the year ended December 31, 20x4. Budgeted Actual Direct labor-hours 75,000 dlh 77,500 dlh Factory overhead $525,000 $558,000 Assume that...
-
If 4.0 g of steam at 100.0°C condenses to water on a burn victim's skin and cools to 45.0° C, (a) how much heat is given up by the steam? (b) If the skin was originally at 37.0°C, how...
-
The authors give whom the credit for being the first forensic accountant? a. James McCleland b. Doug Carmichael c. Maurice E. Peloubet d. Joe Wells e. Robert Lindquist
-
Lang Corporation makes and sells state-of-the-art electronics products. One of its segments produces The Math Machine, an inexpensive calculator. The companys chief accountant recently prepared the...
-
A. The following are some of the elements which form part of the consolidated statement of financial position: a. Investment b. Intra-group Assets and Liabilities C. Intra-Group dividends (if any) d....
-
A mixture of chloroform (CHC13) and acetic acid at 18oC and 1 atm (101.3 kPa) is to be extracted with water to recover the acid.(a) Forty-five kilograms of a mixture containing 35 wt% CHC13 and 65...
-
How do policy decisions affect the development of energy technologies?
-
Describe some advantages and disadvantages of the three types of fossil fuels.
-
Modify your code from the previous exercise so that it could easily be modified to display a different range of numbers (instead of 1234567890 ) and a different number of repetitions of those numbers...
-
Which inventory costing method is frequently used when undifferentiated units are stored in a common area? a. Specific identification b. Weighted-average cost c. FIFO d. LIFO
-
Which inventory costing method does not require the use of the lower-of-cost-or-net realizable value method? a. Specific identification b. Weighted-average cost c. FIFO d. All methods require the use...
-
To what extent is the hypothesized usefulness of flowcharts supported by empirical research results?
-
Which of the following concepts relates to the elimination or minimization of inventories by a manufacturing firm? a. Quick response b. Just-in-time c. Just-in-case d. Specific identification
-
Which inventory costing method assumes that the most recently purchased merchandise is sold first? a. Specific identification b. Weighted-average cost c. FIFO d. LIFO
-
Banks often record transactions on an account in order of the times of the transactions, but many people like to receive their bank statements with checks listed in order by check number. People...
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
N-Phenylsydnone, so-named because it was first studied at the University of Sydney Australia, behaves like a typical aromatic molecule. Explain, using the H?ckel 4n + 2 rule. :. N-Phenylsydnone
-
1-Phenyl-2-butene has an ultraviolet absorption at max = 208 nm ( = 8000). On treatment with a small amount of strong acid, isomerization occurs and a new substance with max = 250 nm ( = 15,800) is...
-
What is the structure of a hydrocarbon that has M + = 120 in its mass spectrum and has the following 1 H NMR spectrum? 7.25 (5 H, broad singlet); 2.90 (1 H, septet, J = 7 Hz); 1.22 (6 H, doublet,...
-
The fixed utilities cost per month for Barkoff is: Show answer choices None of the answer is correct. $4,040 $4,760 $4,400 $5,100 Question 6 0/2 Barkoff Enterprises, which uses the high-low method to...
-
Acquisition costs such as the fees of accountants and lawyers that were necessary to negotiate and consummate the purchase are O expensed in the period of the purchase O recorded as a deferred asset...
-
27 A1 CORPORATION, INC. Income Statement For the Year Ended December 31, 2026 Net Sales $240 Cost of Goods Sold 150 Gross Profit 90 Operating Expenses 65 $25 Net Income Using vertical analysis, what...

Study smarter with the SolutionInn App


