Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the
Question:
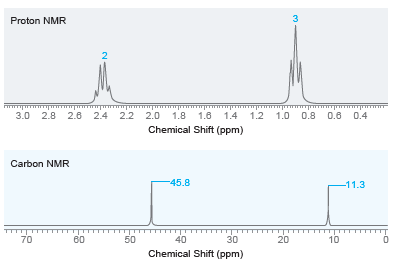
Transcribed Image Text:
3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4 Chemical Shift (ppm) Carbon NMR -45.8 -11.3 40 10 50 70 80 30 20 Chemical Shift (ppm) 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Perhaps more surprising to Mr. Pitkin was a proposal by the VP of Marketing to make a major investment in market share by increasing promotional expenditures by $2.5 million during 1998-2000. Sales...
-
Without calculating, determine whether the value of nPr is greater than the value of nCr for the values of n and r given in the table. Complete the table using yes (Y) or no (N). Is the value of nPr...
-
A material has a strength coefficient K = 100,000 psi Assuming that a tensile-test specimen made from this material begins to neck at a true strain of 0.17, show that the ultimate tensile strength of...
-
What kinds of foreign exchange risk would a U.S. company face that is exporting goods to Mexico? What kinds of things would you monitor to determine what will happen to the value of the Mexican peso?
-
Richmond Company engaged in the following transactions during 2011: a. Purchased $16,000 of supplies from ABC Supplies on February 16. Amount due in full on March 31. b. Paid for 25 percent of the...
-
If F(x) = f(xf(xf(x))), where f(1) = 3, f(3) = 6, f'(1) = 4, f'(3) = 5, and f'(6) = 6. Find F' F'(1) =
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Propose a mechanism for the following process: REN heat + N2 + CO2
-
1. Was Linderman Industries adoption of project organization an appropriate one for getting the Mexican subsidiary started? 2. In consideration of Robert Lindermans letting the division managers know...
-
Which object resoutce typically has the most complex action privileges applying to its use? Briefly explain why this is the case.
-
What is meant by an access control? Why are access controls needed in most computer systems? Can you think of any computer systems that might purposely decrease the level of access control they...
-
Bull and Bear Ltd. is a new, aggressive, Boston-based, medium-sized brokerage firm. It specializes in offering high-quality, personalized service to clients who have a relatively high level of...
-
Briefly explain the nature of the simple security property and the confinement property associated with access controls under a mandatory access control policy.
-
Why is it important that object resources be identified uniquely in a computer system and that the identity of each object resource cannot be forged?
-
Cuomo Mining Corporation, a public company whose stock trades on the Toronto Stock Exchange, uses IFRS. The vice-president of finance has asked you, the assistant controller, to prepare the companys...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
Presented below is information related to Aquinas Co.: Retained earnings, December 31, 2021 $ 750,000 Sales revenue 1,405,000 Additional paid-in-capital 220,000 Loss on sale of equipment 18,000...
-
URGENT Concord Industries can produce 600 units of a necessary component part with the following costs:irect Materials $74100 Direct Labour19800 Variable Overhead 59500 Fixed Overhead9400 If Concord...
-
Corporation A has a profit of $100,000, and Corporation B has a loss of $250,000. Both corporations have the same shareholders. How might Corporation A get the use of Corporation B's losses? a....

Study smarter with the SolutionInn App


