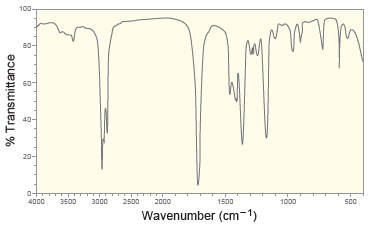
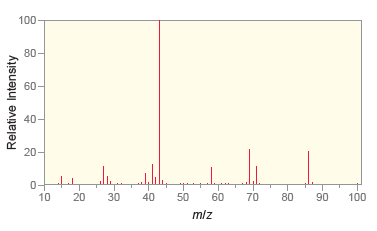
Following are the IR spectrum and mass spectrum of an unknown compound. Propose at least two possible
Question:
Transcribed Image Text:
100- 80- 40- 20- 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) % Transmittance 100 80- 60- 40- 20- 10 20 30 40 50 60 70 80 90 100 mlz Relative Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The IR spetrum indicates that the compound is a ketone T...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose at least two different structures for a compound with six carbon atoms that exhibits the following features: a. All six carbon atoms are sp 2 hybridized. b. Only one carbon atom is sp...
-
Propose at least two strategies to avoid assumptions in a multiyear plan. Justify your response. Recommend at least two best practices for analyzing multiyear financial statements. Justify your...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
In Exercises 7192, find and simplify the difference quotient f(x +h)-f(x) h -, h = 0
-
Define the following: 1. Select database management system (Oracle, SQL Server, MYSQL, etc) and identify the data types and sizes for all attributes. 2. Make sure all relationships have been...
-
Deere & Co. manufactures and distributes farm and construction machinery that it sells around the world. In addition to its manufacturing operations, Deere & Co.'s credit division loans money to...
-
From a lessee's standpoint, leases are classified as finance or operating leases. What criteria are used to identify a finance lease?
-
Knockoffs Unlimited, a nationwide distributor of low-cost imitation designer necklaces, has an exclusive franchise on the distribution of the necklaces, and sales have grown so rapidly over the past...
-
Global Bell is preparing the 2020 year-end information slips for their employees. Joanne had worked for the company in their Montreal, Qubec office from October 1987 until her employment was...
-
Montreal Electronics Company manufactures two large-screen television models: the Nova, which has been produced for 10 years and sells for $900, and the Royal, a new model introduced in early 20x0,...
-
Calculate the HDI for each molecular formula. a) C 4 H 6 b) C 5 H 8 c) C 40 H 78 d) C 72 H 74 e) C 6 H 6 O 2 f) C 7 H 9 NO 2 g) C 8 H 10 N 2 O h) C 5 H 7 Cl 3 i) C 6 H 5 Br j) C 6 H 12 O 6
-
Following are the IR spectrum and mass spectrum of an unknown compound. Propose at least two possible structures for the unknown compound. 100 80 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber...
-
Why do effective speakers moderate their gestures?
-
Wildhorse Company's accounting records reflect the following inventories: Dec. 31, 2022 Dec. 31, 2021 Raw materials inventory $240000 $190000 Work in process inventory 300000 160000 Finished goods...
-
Pool your time length data with data from other class members or the entire class so that the pooled data set has at least 100 observations. Designate someone in the group to calculate the mean and...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
Roger set his residence on fire for the insurance. While the fire was being fought, the police arrived to help with the crowds of onlookers. Roger realized he had done something wrong and went to one...
-
1. a. At the end of ten years how much is an initial Tk. 20,000 deposit followed by five each year-end annual Tk. 5,000 payments worth at the compound interest rate of (6+ last digit of your ID) %...
-
Exercise 1 involves 8 randomly selected deaths during the time period spanning from the week before Thanksgiving to the week after Thanksgiving. Assume now that 20 deaths are randomly selected from...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Into the same funnel is poured carefully 50 mL of hexane (density = 0.660 g/ml) so that the other two layers are not disturbed. The hexane forms a third layer. The funnel is stoppered and the mixture...
-
Give a general balanced reaction for The complete combustion of a cycloalkane containing one ring formula CnH2n.
-
Carv and Di Oxhide drive their family car about12,000 miles per year. Their car gets about 25 miles per gallon of gasoline. "What is the carbon footprint" (pounds of CO, released into the atmosphere)...
-
The second step in the strategic-management process is to look at where the organization stands and see what is working and what could be different to maximize efficiency and effectiveness in...
-
The ending balance of the retained earnings account appears in a.both the statement of stockholders' equity and the balance sheet b.both the statement of stockholders' equity and the statement of...
-
The following items are taken from the financial statements of Tracy Company for 2022. Tracey Company will prepare a balance sheet at December 31, 2022 and an income statement from January 1, 2022 to...

Study smarter with the SolutionInn App


