For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic,
Question:
(a)
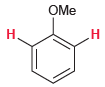
(b)
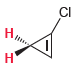
(c)
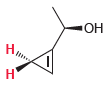
(d)
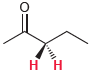
(e)
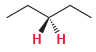
Transcribed Image Text:
OMe Н. Н CI H,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a Homotopic b ...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
Give two names for each of the following compounds, and tell whether each alkyl halide is primary, secondary, or tertiary: a. b. c. d. CH3CH2CHCH3 Cl CHCHCHCHCHCHCl - - - CH3 Br CH CHCH
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
Neutrons can be used in diffraction experiments to probe the lattice structure of crystalline solids. Since the neutron's wavelength needs to be on the order of the spacing between atoms in the...
-
What are the differences between brokered and dealer markets? Would the New York Stock Exchange fit into one of these categories?
-
Calculate the pressure of O2 (in atm) over a sample of NiO at 25C if G = 212 kJ/mol for the reaction NiO(s) Ni(s) 1/2 O2(g)
-
If accounts receivables increase by \($15,000\) and accounts payables increase by \($11,000,\) the effect on cash flow is a. Increase of $26,000 b. Increase of $4,000 c. Decrease of $4,000 d....
-
Bunker makes two types of briefcase, fabric and leather. The company is currently using a traditional costing system with labor hours as the cost driver but is considering switching to an...
-
Cabinor, Inc., is an incinerator construction company. It operates by constructing incinerators for corporations or by providing incinerator management services to local governments. On January 1,...
-
Calculate the number of salespeople required to cover the market. in the present setup (use Table 2) [4 marks]b. with the proposed new setup (use Table 3) [4marks] Type of Shops Pan Shops Kirana...
-
Suppose that a price discriminating monopolist has segregated its market into two groups of buyers. The first group described by the demand and revenue data that you developed for problem 1. The...
-
Butane (C 4 H 10 ) exhibits only two different kinds of protons, shown here in red and blue. (a) Explain why all four protons shown in red are chemically equivalent. (b) Explain why all six protons...
-
This exercise continues our accounting for Grahams Yard Care Inc. from previous chapters. In this exercise, we will account for the annual depreciation expense for Grahams Yard Care, Inc. In the...
-
Would like to understand where is the allocation base, " labour-hour rate "? The question said we have to use labour-hour rate as allocation base. Isn't it different from labour-hour? Why the answer...
-
Question 2- Dynamic odds-and-evens Problem Definition Write a program called odds_evens01 that reads a (non-empty) sequence of integers. As for question 1 above, your program should first ask for the...
-
Consider the situation when the expense is required on purchasers. In contrast to when assessment is forced on makers, the interest bend movements to one side to make new harmony with beginning...
-
As a financial manager you believe that a 20 percent rate of return is reasonable given the nature of the risks involved in your company's $120,000 investment. Your company will receive $48,000 in...
-
A warehouse receives 15,000-unit loads, on average, per day. Each unit load is stored for 1 month (30 days) in storage, on average. The space required for a unit load is 1 cubic meter (1 m' ). What...
-
Ms. Jobe has been very ill since the beginning of the year and unable to attend to any financial matters. Her CPA advised that she request an automatic extension of time to file her prior year Form...
-
Refer to the data in QS 10-1. Based on financial considerations alone, should Helix accept this order at the special price? Explain.
-
Locate and identify the functional groups in the following molecules. In these representations, each intersection of lines and the end of each line represents a carbon atom with the appropriate...
-
Draw structures that meet the following description (there are many possibilities): (a) Three isomers with the formula C8H18 (b) Three isomers with the formula C4H8O2
-
Draw structures of the nine isomers of C7H16.
-
Cathy Company reported $500 in wage expense but did not report any wages payable on its beginning or ending balance sheets. Cash paid for wages is: $0 more than $0, but less than $500. more than...
-
Micro cost/benefit analysis In 1984, Congress passed the National Organ Transplant Act, which outlawed the buying and selling of internal organs. The National Task Force on Organ Transplantation...
-
Labeau Products, Limited, of Perth, Australia, has $34,000 to invest. The company is trying to decide between two alternative uses for the funds as follows: Invest in Project X Invest in Project Y...

Study smarter with the SolutionInn App


