Identify the major and minor products for the E2 reaction that occurs when each of the following
Question:
a.
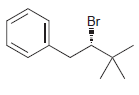
b.
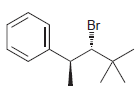
c.
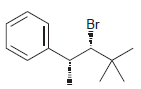
d.
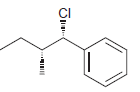
e.
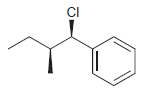
f.
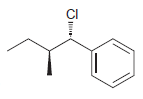
g.
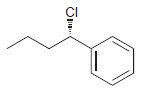
h.
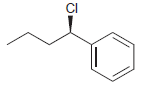
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
a b c...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When menthyl chloride is treated with a strong base, only one elimination product is observed. Yet, when neomenthyl chloride is treated with a strong base, two elimination products are observed. Draw...
-
When (1R, 2R)-2-bromocyclohexanol is treated with a strong base, an epoxide (cyclic ether) is formed. Suggest a mechanism for formation of the epoxide: Strong base Br An epoxide
-
Identify the major and minor products for each of the following E2 reactions: a. b. c. d. e. f. CI NaOEt CI t-BUOK
-
. What is the output of the following program? Write your answer in the box below. #include using namespace std; int main() { int a=4; int b=6; int i; int j; } int** k; i = &a; j = &b; *j = 8; if...
-
A sample of soil plus container weighs 397.6 g when the initial water content is 6.3%. The container weighs 258.7 g. How much water needs to be added to the original specimen if the water content is...
-
Consider the graph of y = x2. Let matrix [P], which organizes the coordinates of the points shown, represent five points on the parabola. a. Describe the transformation(s) that would give five...
-
The net income for Robin Williams Co. for 2002 was \($250,000\). For 2002, depreciation on plant assets was \($60,000\), and the company incurred a loss on sale of plant assets of \($10,000\)....
-
Briggs & Stratton is the worlds largest producer of air-cooled gasoline engines for outdoor power equipment. Its 2011 annual report to stockholders included the following data (in millions): Net...
-
Delta Company produces a single product. The cost of producing and selling a single unit of this product at the companys normal activity level of 94,800 units per year is: Direct materials $ 1.60...
-
Dexter, Inc., had a cost of goods sold of $75,318. At the end of the year, the accounts payable balance was $18,452. How long on average did it take the company to pay off its suppliers during the...
-
Show two different methods for preparing each of the following alkenes (one method using a sterically hindered base and the other method using a base that is not sterically hindered): a. b.
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
A spherical convex mirror has a radius of curvature with a magnitude of 40.0 cm. Determine the position of the virtual image and the magnification for object distances of (a) 30.0 cm and (b) 60.0 cm....
-
The owners of the Wilson Building desire to refinance their building.They seek the following loan terms: LOAN REQUEST: Amount:$7,500,000 Rate:6.0% Term:10 years Amortization: 30 years QUESTIONS: (The...
-
Taxpayer X filed a 2021 return on February 21, 2022, showing a tax liability of $8,700. On August 15, 2022, he filed an amended return showing an additional tax liability of $737 (he found a couple...
-
Fill in the gap: $customers ); 'name' => $name, 'id' => $id = array (
-
48 Which organization maintains the payroll master data for a client and performs payroll processing? Opal employer organization Orched distribution organization
-
Though several types of control activities are needed in an effective system of internal controls, which type is generally considered the most effective? O Separation of duties O Proper authorization...
-
Refer to Exercise 91. a. How do the ranges of the two distributions compare? Justify your answer. b. Explain why the standard deviation of the total family incomes in the New Jersey sample is so much...
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
How would you prepare ds-2-butene starting from propyne, an alkyl halide, and any other reagents needed? This problem cant be worked in a single step. Youll have to carry out more than one reaction.
-
Beginning with 4-octyne as your only source of carbon, and using any inorganic reagents necessary, how would you synthesize the following compounds? (a) cis-4-Octene (b) Butanal (c) 4-Bromooctane (d)...
-
Beginning with acetylene and any alkyl halides needed, how would you synthesize the following compounds? (a) Decane (b) 2, 2-Dimethylhexane (c) Hexanal (d) 2-Heptanone
-
Oregon can not tax interest or dividends on obligations that states cannot tax under federal law. Examples of such obligations include which of the following? Question 7Select one: a. Bonds issued by...
-
Which of the following defines long-term liabilities? Multiple choice question. Long-term liabilities are reported before current liabilities on a classified balance sheet. Long-term liabilities are...
-
Which of the following individuals has properly disclosed rates and terms related to a Refund Anticipation Loan (RAL)? Question 15Select one: a. Tax Consultant Jerry has verbally discussed with his...

Study smarter with the SolutionInn App


