Kodel is a synthetic polyester with the following structure. (a) Identify what monomers you would use to
Question:
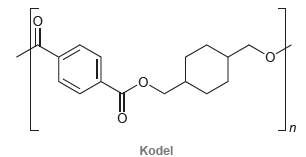
(a) Identify what monomers you would use to make Kodel.
(b) Would you use acidic conditions or basic conditions for this polymerization process?
Transcribed Image Text:
in Kodel
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a b Ac...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Kodel is a polyester with the following structure: From what two monomers is it made? -O-CH CH2-O
-
The compound benzenc has only one type of carbon-carbon bond, and this bond has a length intermediate between that of a single bond and a double bond. Draw a resonance structure of benzene that,...
-
Amino acids are biological compounds with the following structure, where the R group can vary. The structure and biological function of amino acids will be discussed in Chapter 25. Identify the total...
-
Use the present value tables in Appendix A and Appendix B to compute the NPV of each of the following cash inflows. a. $89,000 received at the end of six years. The discount rate is 4 percent. b....
-
True Fit Shoe Company makes loafers. During the most recent year, True Fit incurred total manufacturing costs of $21,900,000. Of this amount, $2,600,000 was direct materials used and $14,800,000 was...
-
Anna does not make quarterly estimated tax payments even though she has substantial amounts of income that are not subject to withholding. In 2012, Anna's tax liability was $18,000. In 2013, Anna's...
-
Net income was \($500,000\) in 2000, \($420,000\) in 2001, and \($504,000\) in 2002. What is the percentage of change from (a) 2000 to 2001 and (b) 2001 to 2002? Is the change an increase or a...
-
On paper, replicate the following segment of DNA: 5 A T C G G C T A C G T T C A C 3 3 T A G C C G A T G C A A G T G 5 a. Show the direction of replication of the new strands and indicate the location...
-
Flounder Company, a manufacturer of audio systems, started its production in October 2022. For the preceding 3 years, Flounderhad been a retailer of audio systems. After a thorough survey of audio...
-
Aberdeen Computer Corp. (ACC) is located in Aberdeen, Washington. The company has developed the Web-Surfer, a low-cost e-mail and Web-surfing appliance. This product is manufactured at four plants,...
-
Explain why vinyl shower curtains develop cracks over time.
-
Polyformaldehyde, sold under the trade name Delrin, is a strong polymer used in the manufacture of many guitar picks. It is prepared via the acid-catalyzed polymerization of formaldehyde. (a) Draw...
-
In Problems 7984, graph each function using a graphing utility and the Change-of-Base Formula. y = logx+2(x - 2)
-
Beverly Company has determined a standard variable overhead rate of $3.90 per direct labor hour and expects to incur 0.50 labor hours per unit produced. Last month, Beverly incurred 1,650 actual...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $25,760 of office salaries and $60,840 of sales salaries. Withholdings from the employees' salaries...
-
Let be an alphabet. The symmetric difference of two languages A, B C * is defined as AAB = (ANB) U (ANB). (a) Prove that if A C* is a context-free language and BC * is a finite language, the the...
-
The pressure relief valve in your hot water heater (sizedesignation: Type G) has an orifice area of 0.503 in2 . It is setto open and relieve the pressure in the heater if it 23 reaches 150psi. The...
-
3- Imagine a system that has n processes and a resource with m identical units. The maximum claim of any process is k. a. Why is the system deadlock free for all possible states of m>n (k-1)? (1...
-
Repeat Exercise 7.8 for the multicycle processor. Assume the instruction mix of Example 7.7.
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitro compounds and the Gabriel synthesis leave the carbon chain unchanged. Formation...
-
(a) Guanidine (shown) is about as strong a base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines. (b) Show why p-nitroaniline is a much weaker base (3 pKb units...
-
Show how you would synthesize the following compounds starting with benzene, toluene, and alcohols containing no more than four carbon atoms as your organic starting materials. Assume that para is...
-
Question 4 of 6 < > -/15 Current Attempt in Progress Sheridan Company has accounts receivable of $202,000 at September 30, 2024. An analysis of the accounts shows the following: Month of Sale Balance...
-
The charge on your rent-to-own washer is $40 per month. After 32 months, you get to keep the washer. How much does the washing machine end up costing you? Cost of washing machine
-
Prepare horizontal analysis on income statement below: Bristol Myers Squibb Co. (NYS: BMY) Report Date 2020 2019 2018 Net product sales 41,321 25,174 21,581 Alliance & other revenues 1,197 971 980...

Study smarter with the SolutionInn App


