Predict the major product for each E1 reaction: a. b. eat Br H2SO4 eat
Question:
a.
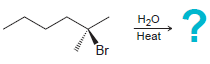
b.
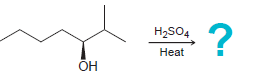
Transcribed Image Text:
Нао Нeat Br H2SO4 Нeat ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a b ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
Predict the major product for each of the following E2 reactions: a. b. c. d. NaOH Br NaOH Br
-
Predict the major product for each of the following reactions: a. b. c. d. e. f. g. h. i. j. CI :? NaSH DBN OTs
-
1. [20 points] A mattress manufacturer has three production facilities located in Pittsburg (PA), Houston (TX), and Los Angeles (CA). They supply their products to three distribution centers in...
-
To be effective, a company's public relations efforts should be integrated with advertising efforts in the marketing mix and should be directed to achieving the same promotional goals. We do this by...
-
How can you find the horizontal asymptote of a rational function without graphing or using a table? Use a calculator to explore these functions, and look for patterns. Some equations may not have...
-
Barrila Enterprises uses a word processing computer to handle its sales invoices. Lately, business has been so good that it takes an extra 3 hours per night, plus ever} third Saturday, to keep up...
-
Gonzales, CPA, is the auditor for a manufacturing company with a balance sheet that includes the entry Property, plant, and equipment. Gonzales has been asked by the companys management if audit...
-
10. Seghen's car was destroyed by a tornado in a Federally declared disaster area in the current year. At the time of the accident, the car had a fair market value and an adjusted basis of $30,000....
-
Digby's product manager is considering lowering the price of the Drat product by $2.50 and wants to know what the impact will be on the product's contribution margin . Assuming no inventory carry...
-
You are a newspaper publisher. You are in the middle of a one-year rental contract for your factory that requires you to pay $500,000 per month, and you have contractual labor obligations of $1...
-
One advantage of the various forms of renewable energy, such as solar thermal and wind energy is that they cause no net increase in atmospheric carbon dioxide. Is this true for biomass? Why or why...
-
Why is it important to separate content from display when designing an Internet-based electronic commerce system?
-
One of the distinguishing characteristics of IT firms is that their assets are mainly composed of intangible assets such as patents, software, research and development potential, innovation capacity...
-
Achievable Company produces component A1 for use in one of its electronic gadgets. Normal annual production for the item is 100,000 units. The cost for a 100-unit batch of the part are: Direct...
-
A company's issued share capital consists of $ 2 0 0 , 0 0 0 in 5 0 c ordinary shares issued at a premium of 1 0 c per share. A dividend of 2 0 % is declared. what is the cash payable to shareholders...
-
Recently, Shasta Corporation has decided to play a more-active role in the soda be is aware that the current market price for a can of soda is $2.00. Shasta plans to se soda in its first year and...
-
In the 2nd stage regression used to test the CAPM, researchers reduced the measurement error in betas from the 1ststage regression. What was the source of the measurement error and how did they...
-
For the piled raft in Problem 14.2, if K r = 3250 MN/m and K p = 5300 MN/m, find K pr and X, the fraction of the load carried by the raft. Problem 14.2 In a piled raft, 450 mm diameter and 13 m long...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
The Darzens reaction involves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second...
-
The following reaction involves a hydrolysis followed by an intra molecular nucleophilic acyl substitution reaction. Write both steps, and show theirmechanisms. CH CH H30* CHCH CH2CO2H
-
The following reaction involves an intra molecular Michael reaction followed by an intra molecular aldol reaction. Write both steps, and show theirmechanisms. NaOH Ethanol
-
on The following details were obtained from the books of Tia Maria Ltd: 200,000 ordinary shares @ $0.50 were repurchased at par value o Interest charges of $75,000 was paid during the year o...
-
Transaction 1 On March 1, the three classmates opened a checking account for The Wire at a local bank. They each deposited $20,000 in exchange for shares of stock. A few of their friends also...
-
At January 1, 2022, Marigold, Inc. has beginning inventory of 9400 boogie boards. Marigold estimates it will sell 6000 units during the first quarter of 2022 with a 6% increase in sales each quarter....

Study smarter with the SolutionInn App


