Predict the product and draw the mechanism for each of the following reactions: a. b. c. 1)
Question:
a.
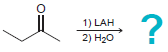
b.
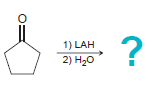
c.
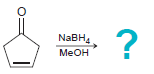
Transcribed Image Text:
1) LAH 2) H20 1) LAH 2) H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a b c ...View the full answer

Answered By

ABHISHEK S
I started teaching when I was studying at college .I taught some of my juniors in college.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following reactions, predict the product and draw the mechanism of its formation. a. b. c. d. e. f. 1) PhMgBr 2) H20 Me 1) NaCN 2) H20 *Me
-
For each reaction, predict the product and draw the mechanism of its formation. a. b.
-
Draw a mechanism for each of the following E1 processes: a. b. c. d. H,SO, Heat Br ETOH, Heat
-
If you can't find similar ratios to class-covered ones, use financial formulas to calculate them manually. Choose up to two of the following: Operating Margin EBITDA Margin Payout Ratio 3. Analysis...
-
Selected data from Komar Company follow. a. Compute the accounts receivable turnover for 2014. (Round your answer to 2 decimal places Accounts receivable turnover: times b. Compute the inventory...
-
Telescopes used in astronomy have large lenses (or mirrors). One reason is to let a lot of light in important for seeing faint astronomical bodies. Can you think of another reason why it is an...
-
5. Until recently, every state had a statute outlawing the burning of American flags. But in Texas v. Johnson, the Supreme Court declared such statutes unconstitutional, saying that flag burning is...
-
Everly Corporation acquires a coal mine at a cost of $400,000. Intangible development costs total $100,000. After extraction has occurred, Everly must restore the property (estimated fair value of...
-
The fiscal year-end unadjusted trial balance for Nelson Companyis found on the trial balance tab. Rent expense and salariesexpense are equally divided between selling activities and thegeneral and 2...
-
Derive the transfer function H(s)/Q(s) for the liquid-level system of Fig. P5-1 when (a) The tank level operates about the steady-state value of hs = 1 ft (b) The tank level operates about the...
-
A bottle at 325 K contains an ideal gas at a pressure of 162.5 10 3 Pa. The rubber stopper closing the bottle is removed. The gas expands adiabatically against P external = 120.0 10 3 Pa, and some...
-
Predict the product and draw the mechanism for each of the following reactions: a. b. c. ? 1) LAH 2) H20
-
The following shows ball-and-stick models of the reactants in a Lewis acidbase reaction. Write the complete equation for the reaction, including the product. Identify each reactant as a Lewis acid or...
-
ucational s using a non-rebreather face mask? Assume the tank is 2000 psi ucational Grou 31. What is the estimated duration of oxygen supply from a D-size oxygen tank, delivering oxygen at a rate of...
-
This case focuses on developing a deeper understanding of the personnel scheduling problem. Big Town Fire Department would like to schedule their human resource requirements in fire and safety...
-
If you have a MongoDB source feeding into your data warehouse and an individual field value changes, what is the downstream impact? What is the downstream impact if the schema of new documents in...
-
After General Mills experienced such backlash about its new legal terms, the company issued a statement reverting back to the original policy. Imagine that you're the head of corporate communications...
-
Despite its advantageous position and relatively low labor costs, outside of automotive Mexico has not received nearly as much offshoring from US as China. Why? What specific steps can Mexico take to...
-
Explain why the vertical-line test used to identify functions in rectangular coordinates does not work for equations expressed in polar coordinates.
-
True & False The basis of an asset must be reduced by the depreciation allowable, 2. Adjusted gross income (AGI) is the basis for a number of phase-outs of deductions. 3. A change to adjusted gross...
-
Propose a structure for an octapeptide that shows the composition Asp, Gly2 Leu, Phe, Pro2, Val on amino acid analysis. Edman analysis shows a glycine N-terminal group, and leucine is the C-terminal...
-
The reaction of ninhydrin with an ?-amino acid occurs in several steps. (a) The first step is formation of an imine by reaction of the amino acid with ninhydrin. Show its structure and the mechanism...
-
Draw resonance forms for the purple anion obtained by reaction of ninhydrin with an a-amino acid.
-
_____ are organizations, typically financed by a combination of government tax revenues and private sector funds, whose primary roles are to help bring tourists into an area and to generate income in...
-
Suppose all of the major furniture producers announced that beginning next month there would be major price increase of furniture. This would cause the current demand for furniture to: a decrease. b...
-
In a market economy, Answers A. people trade goods directly through the barter system. B. private businesses sell products, and consumers buy products. C. people grow and produce all the food and...

Study smarter with the SolutionInn App


