Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b)
Question:
(a)
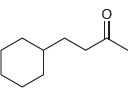
(b)
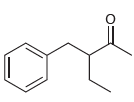
(c)
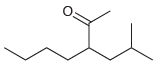
(d)
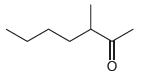
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a b c d ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH H.
-
Propose an efficient synthesis for each of the following transformations: a. b. c. d. Br
-
Describe two methods for determining the molecular mass of a polypeptide. Which is more accurate and why?
-
An independent polling organization interviewed 100 college students to determine their political party affiliations and whether they favor a balanced-budget amendment to the Constitution. The table...
-
Let X(t) be a Brownian motion process with variance Var[X(t)] = t. For a constant c > 0, determine whether Y(t) = X(ct) is a Brownian motion process.
-
What role does MIS play? Do they have a chief information officer? Do they have a Web site? How in-depth do you consider it to be? You may wish to compare it with other sites.
-
Oceanview School, a private high school, is preparing a planned income statement for the coming academic year ending August 31, 2013. Tuition revenues for the past two years ending August 31 were as...
-
Sketch a triangle that has acute angle e tante NIM S Find the other five trigonometric ratios of SED U 13 2 3
-
The following accounts are taken from the records of Jasper Inc. at January 31, 2015, its first month of operations. Required: 1. Calculate the amount of total assets. 2. Calculate the amount of...
-
In Problem 22.38, we saw an intramolecular example of a malonic ester synthesis using excess base and 1,4-dibromobutane. If this dibromide is used in an acetoacetic ester synthesis, an intramolecular...
-
Guanidine is a neutral compound but is an extremely powerful base. In fact, it is almost as strong a base as a hydroxide ion. Identify which nitrogen atom in guanidine is so basic, and explain why...
-
Select the best answer for each of the following questions and explain the reasons for your choice. a. If a CPA recklessly performs an audit, the CPA will be liable to third parties who were unknown...
-
Describe why teams are being formed more frequently in business today. What are some of the special considerations required for a team to be successful?
-
_____ is providing a reward in return for a constructive action on the part of the subject.
-
Describe a situation where empowerment has been used. What are the benefits and drawbacks to empowerment in the situation?
-
Briefly explain the limitations of natural-language interfaces to an application system with respect to: a. Ambiguity of commands b. Establishing the lexicon c. Ambiguity in responses d. Changes to...
-
Developing countries have complained that the terms of trade they face are unfavorable. If they voluntarily engage in international trade, what do you suppose they mean by unfavorable terms of trade?
-
On September 1, 2020, Wong Corporation, which uses ASPE, signed a five-year, non-cancellable lease for a piece of equipment. The terms of the lease called for Wong to make annual payments of $13,668...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
In each pair of compounds, which compound has the higher boiling point? Explain your reasoning. (a) octane or 2,2,3-trimethylpentane (b) Nonane or 2-methylheptane (c) 2, 2, 5-trimethylhexane or nonane
-
Angela is entitled to a fixed annual payment of $15,000 from ABC Partnership without regard to the income of the partnership. Her share of the partnership income is 10%. After deducting the...
-
Rosie has $24,000 short-term capital loss, $10,500 28% gain and $9,000 0%/15%/20% gain. Which of the following statements is correct? a. Rosie has a $19,500 net capital gain b. Rosie has a $7,500...
-
Wyatt Company was formed on January 1, Year 1, when it acquired $50,000 cash from issuing common stock. Which of the following shows the impact of this transaction on Wyatt's accounting equation?...

Study smarter with the SolutionInn App


