Suggest an efficient synthesis for each of the following transformations: a. b.
Question:
a.
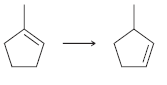
b.
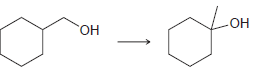
Transcribed Image Text:
Он ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (17 reviews)
a b 1...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) (g) -OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b)
-
(a) Find the Laplace transform of the voltage shown in Fig. 16.52(a). (b) Using that value of vs (t) in the circuit shown in Fig. 16.52(b), find the value of v0 (t). v,(0) 3 V 0 1s al 112
-
identify the theory that underlies the proverb or rule of thumb, and generate a testable hypothesis, and suggest a possible strategy (descriptive, correlation, or experimental) to test the...
-
Let f (x) = x2/3. Find f (S) if a) S = {2,1, 0, 1, 2, 3}. b) S = {0, 1, 2, 3, 4, 5}. c) S = {1, 5, 7, 11}. d) S = {2, 6, 10, 14}.
-
Calculate the amount recorded as the cost of a new machine given the following payments related to its purchase: gross purchase price, $700,000; sales tax, $49,000; purchase discount taken, $21,000;...
-
Global Cell Phones (GCP) has developed a cell phone that can be used anywhere in the world (even countries like Japan that have a relatively unique cell phone system). GCP has been receiving...
-
QUESTION ONE Based on your knowledge of the conceptual framework for Financial reporting, identify and justify the accounting concept/principle applicable to the situations described below: (10...
-
A system consisting of 1 kg of H2O undergoes a power cycle composed of the following processes: Process 1-2: Constant-pressure heating at 10 bar from saturated vapor Process 2-3: Constant-volume...
-
Looking at the Figure 32.3, maintaining P floor as the target minimum price (rather than equilibrium) by having the government purchase how much of the product farmers wished to produce would cost...
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 2-bromobutane.
-
Describe the regulatory structure for monitoring audit quality in the UK.
-
Huang Automotive is presently operating at 75% of capacity. The company recently received an offer from a Korean truck manufacturer to purchase 25,500 units of a power steering system component for...
-
What is the management accounting information needed of the manager of a patient unit where patients stay while being treated for illness or while recuperate from an operation?
-
(1) Why accrual basis accounting may create the overproduction incentive in the absorption costing but not in the variable costing method? (2) As an accountant, what are your recommendations to the...
-
A fixed asset with a cost of $27,412 and accumulated depreciation of $24,670.80 is sold for $4,660.04. What is the amount of the gain or loss on disposal of the fixed asset?
-
Section 1 8 0 requires directors to act with minimum standards of care, skill, and diligence; this is expected of all directors. Whilst directors need to be bold and to be able to manage their...
-
Based on Schrage (1997). The file P08_05.xlsx lists the size of the four main markets for Excel, Word, and the bundle of Excel and Word. (We assume that Microsoft is willing to sell Excel or Word...
-
When the concentration of a strong acid is not substantially higher than 1.0 10-7 M, the ionization of water must be taken into account in the calculation of the solution's pH. (a) Derive an...
-
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction. 1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
-
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is...
-
An unknown compound has the formula C6H10. (a) What is the DU for this compound? (b) When a solution of Br2 in CC14 is added to the unknown, the bromine color disappears. What information does this...
-
Sort the following into asset, liability and stockholder equityAccounts payable. . . . . . . Liability Accounts receivable. . . . . Asset Advertising expense. . . . . ? Building. . . . . . . . . . ....
-
In applying the present value rule for a lease classification, the present value of the residual value a. should be included regardless of guaranteed or not. b. should only be included if it is...
-
When it comes to cost allocation, there are various best practices that can be used. One is to thoroughly record the allocation process. This helps to guarantee that costs are allocated consistently...

Study smarter with the SolutionInn App


