The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14
Question:
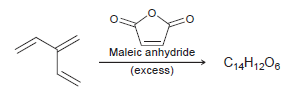
Transcribed Image Text:
Maleic anhydride (excess) C14H1208
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider a process that attempts to prepare tyrosine using a HellVolhardZelinski reaction: (a) Identify the necessary starting carboxylic acid. (b) When treated with Br 2 , the starting carboxylic...
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
A mixture of maleic anhydride and benzoic acid containing 10 mol% acid is a product of the manufacture of phthalic anhydride. The mixture is to be distilled continuously in a column with a total...
-
On September 1, 2025, Swifty Corporation acquired Windsor Enterprises for a cash payment of $800,000. At the time of purchase, Windsor's balance sheet showed assets of $570,000, liabilities of...
-
The state of plane stress in a body is described by the following stresses: 1 = 8500 kN/m2 compression, 3 = 1500 kN/m2 tension. Determine by means of the Mohr circle the normal stress and shear...
-
The purchase price of a natural gas-fired commercial boiler (capacity X) was $181,000 eight years ago. Another boiler of the same basic design, except with capacity 1.42X, is currently being...
-
Mastercraft Inc. makes a variety of motordriven products for the home and small businesses. The market research department recently identified power lawn mowers as a potentially lucrative market. As...
-
At a mine processing site a new process section requires power that will be by an underground service. It will be fed by a 5MVA 22/3.3kV 7.5% Z transformer. (3ph fault power 66.7 MVA, fault current...
-
REQUIRED a. b. C. Prepare a combined adjustment journal entry in the general journal of Sentosa Traders on 30 April 20.5. Show the bank account for April 20.5 in the general ledger of Sentosa...
-
Conan Industries collected $100,000 from customers in 2010. Of the amount collected, $25,000 was from revenue earned on account in 2009. In addition, Conan earned $40,000 of revenue in 2010, which...
-
Government inspectors who check on the quality of services provided by retailers as well as government requirements for licensing in various professions are both attempts to resolve: a. The moral...
-
True or False: A market may collapse and have relatively few transactions between buyers and sellers if buyers have more information than sellers.
-
Simplify. Assume that all variables represent positive real numbers. -V-125abc2
-
10 The balance of finished goods account on December 31 is: * (2 Points) The following balances were taken from the records of ABC Corp., a manufacturer of first-class home decors. January 1, 2015...
-
What are the key components of strategic leadership, and how do visionary leaders effectively articulate and execute a compelling vision, fostering organizational alignment and sustainable...
-
2. Describe the various methods of quality control and quality assurance you will be using to maintain the product's quality through out the assembly process and supply zero-defect quality. What are...
-
6. A 70-year-old surviving widow with good eyesight is not claimed as a dependent on anyone else's tax return. The widow's gross income threshold for purposes of filing a return in 2022 is: a. b. C....
-
Choose EITHER a recent good service experience OR a recent bad service experience. Briefly convey the circumstances of the experience in your initial post. Next, discuss which elements of services...
-
Canada has two official languages, English and French. Choose a Canadian at random and ask, What is your mother tongue? Here is the distribution of responses, combining many separate languages from...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Why do you suppose ketone halogenations in acidic media referred to as being acid-catalyzed, whereas halogenations in basic media are base-promoted? In other words, why is a full equivalent of base...
-
How could you use a malonic ester synthesis to prepare the following compounds? Show allsteps. (b) CH3CH2CH2CHC CH (c) CHCH2C CHCHCH-CH,C C
-
Monoalkylated and dialkylated acetic acids can be prepared by the malonic ester synthesis, but trialkylated acetic acids (R3CCO2H) cant be prepared. Explain
-
Mint, now part of Intuit, entered the market in 2005 with comprehensive online money management systems that by aggregating financial information from online bank and credit card accounts tell users...
-
Which benefit has federal regulations that directly tie the physician's benefit to the employees' benefit (if offered to physician must also be offered to employee)? a. Flexible Spending Account...
-
Which of the following is a prohibited act under Chapter 673? Question 20Select one: a. To use a suspended license b. To prepare a fraudulent return c. To use the license of another person d. All of...

Study smarter with the SolutionInn App


