Using bromobenzene and ethylene oxide as your only sources of carbon, show how you could prepare trans-
Question:
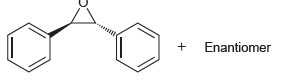
Transcribed Image Text:
+ Enantiomer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Br 1 Mg 2 3 HO MCP...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2-Phenylethanol, which has the aroma of oil of roses, is used in perfumes. Write equations to show how 2-phenylethanol can be synthesized from bromobenzene and ethylene oxide, using a Grignard...
-
Using acetylene and ethylene oxide as your only sources of carbon atoms, propose a synthesis for each of the following compounds. a. b.
-
Design a synthesis of 3-pentyn-1-ol using propyne and ethylene oxide as the only sources of carbon atoms.
-
Explain the purpose, uses and advantages of classified statements of cash flows prepared in accordance with IAS 7.
-
Provide an implementation of a removeAll method for the MyLinkedList class. Method removeAll removes all items in the specified collection given by items from the MyLinkedList. Also provide the...
-
A one tossed vertically into the air with Initial velocity v cm/s reaches a maximum height of h = v2/ 1960 cm. (a) Estimate h if v = 700 cm/s and v = 1 cm/s. (b) Estimate h if v = 1000 cm/s and v = 1...
-
Discuss implicit theories and leadership prototype.
-
Sebastian and Whitley, plumbers, successfully bid $30,000 for the plumbing work on a new luxury home. Total direct labor cost on the job was $9,500, other direct costs were $2,500, and overhead is...
-
at a ids. BE15.4 (LO 2) Nasreen Company issues $2 million, 10-year, 8% bonds at 97, with interest payable each January 1. a. Prepare the journal entry to record the sale of these bonds on January 1,...
-
The inverse market demand in a homogeneous-product Cournot duopoly is P = 100 2(Q1 + Q2) and costs are C1(Q1) = 12Q1 and C2(Q2) = 20Q2. a. Determine the reaction function for each firm. b. Calculate...
-
Which advantage does a typical developing country have in attempting to draw foreign investment? a. Very low wages b. Poor education c. Easy profit repatriation d. Independent central banks
-
The method of detecting sex discrimination most likely to minimize it would be to use a. Simple differences in income between men and women. b. Simple differences in full-time wages for men and...
-
What can we do about lost sales due to poor customer service by outside "contract" sales staff?
-
5 1 points eBook a. Equipment with a book value of $79,500 and an original cost of $167,000 was sold at a loss of $30,000. b. Paid $103,000 cash for a new truck. c. Sold land costing $325,000 for...
-
Company has a recent gross profit history of 60% based on cost Date available from the accounting record for 3 months ended March 31 Inventory - Jan 1 350,000.00 Purchases 3,650,000.00 Sales...
-
What roles do trust and commitment play in business relationships and Why might the trust and commitment theory be insufficient in explaining the performance of these relationships?
-
Vijay's Deliveries purchased a truck on February 1, 2015. The truck cost $40 000, has a useful life of seven years and a residual value of $5 000. It is estimated that over life of the truck, it will...
-
Kai signs a 10-year lease to rent a property. In the first year, he receives $5,000 for the first year\'s rent and $5,000 as rent for the last year of the lease. What amount must Kai include in...
-
A student performs a test of H 0 : = 12 versus H a : 12 at the = 0.05 significance level and gets a P-value of 0.01. The student writes: Because the P-value is small, we reject H 0 . The data...
-
What is the shape of the exponential distribution?
-
The carbocation electrophile in a FriedelCrafts reaction can be generated in ways other than by reaction of an alkyl chloride with A1Cl3. For example, reaction of benzene with 2-methylpropene in the...
-
The N, N, N-trimethyl ammonium group, N (CH3)3, is one of the few groups that is a meta-directing deactivator yet has no electron-withdrawing resonance effect. Explain.
-
The nitroso group, N = O, is one of the few non-halogens that is an ortho- and Para-directing deactivator. Explain by drawing resonance structures of the carbocation intermediates in ortho, Meta and...
-
Why do parent companies disregard the legal boundaries of entities, and report consolidated financial statements as a single economic entity? Select answer from the options below to provide useful...
-
Identify a true statement about merit increases in a small business. Multiple choice question. It is an arrangement whereby an employee receives a prescribed share of the business's profits. It is...
-
Price Company assigns overhead based on machine hours. The Milling Department logs 1,800machine hours and Cutting Department shows 3,000 machine hours for the period. If theoverhead rate is $5 per...
In Mixed Company Communicating In Small Groups And Teams 11th Edition - ISBN: 0197602819 - Free Book

Study smarter with the SolutionInn App


