Would water be a suitable proton source to protonate the following compound? ONa
Question:
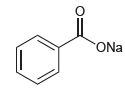
Transcribed Image Text:
ONa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
No water would not be a suitable proto...View the full answer

Answered By

HABIBULLAH HABIBULLAH
I have been tutor on chegg for approx 5 months and had solved a lot of questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We will learn all of the following reactions in upcoming chapters. For each of these reactions, notice that the product is an anion (ignore the positively charged ion in each case). In order to...
-
Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following proton transfer? Explain your answer: ONH2 + NH3 NH2 H.
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Prepare a company analysis (strengths and weaknesses) Instructions - Prepare the SW portion of a SWOT analysis as a report. Use your textbook and the instruction below. Prioritizing the Results: The...
-
Investigate learning management software packages that are available through application service providers. Using the World Wide Web, identify at least two potential sources of such software. What...
-
Evaluate the differences in approaches to labor- management relations between the George W. Bush and Obama administrations?
-
Teresa has been working as an advertising copywriter for five years. She has been saving money and now has $10,000 to invest in real estate. Teresa wants to make sure that her real estate investments...
-
The December cash records of Duffy Insurance follow: Duffys Cash account shows a balance of $17,050 at December 31. On December 31, Duffy Insurance received the following bank statement: Additional...
-
The following data apply to Problems 4 through 10: A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term bond fund, and the third is a money...
-
A news network reported the results of a survey about a controversial court decision. The network initially posted on its website a bar graph of the data similar to the one that follows. Explain how...
-
There are four constitutional isomers with molecular formula C 3 H 9 N. Draw a Lewis structure for each isomer and determine the number of lone pairs on the nitrogen atom in each case?
-
Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH 2) ,* z . Pt
-
Write a Network Architecture Paper, Using either the OSI Reference Model or the TCP/IP Protocol Suite; describe how each layer of the Model/Suite represents the communication flow between...
-
Which of the following deviations from company policy would an operational auditor consider to be the most critical? a. Several late payments to vendors resulted in loss of discounts. b. An employee...
-
If the objective of the audit is to determine that adequate environmental protection and alarm devices are installed and operating, the auditor should a. Review the architect's alarm specification...
-
State the rule on contingent fees.
-
State the rule on confidentiality.
-
Which one of the following is the best objective performance criterion for an electrical engineer? a. Exhibition of creative talents on company projects. b. Project completion within budget...
-
A stent is a cylindrical tube, often made of metal mesh, thats inserted into a blood vessel to overcome a constriction. Its sometimes necessary to heat the stent after insertion to prevent cell...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Show the products of this reaction. How would the composition of the products change if t-BuO ? in t-BuOH were used in place of ethoxide ion in ethanol? ELOH + CH,CH,0
-
All of the stereo isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane have very similar rates of E2 reaction except the following stereo isomer, which reacts about 7000 times more slowly than the...
-
Show the product of thisreaction: Ph Br- - + NaOEt Br EIOH Ph
-
step analysis framework of this textbook. Beginning in Chapter 1 and following each chapter of book, we use the Starbucks Integrative Case to illustrate and apply all of the tools of finand...
-
All of the following insureds purchased a long-term care insurance policy after 2000. Based solely on the information given, who does not own a tax-qualified LTCI policy? (Search Chapter 8) a....
-
Price and cost P2 Q Q Q2 Q ATC MC AVC MR Quantity 3) Refer to Figure 10.14. Suppose the prevailing price is P and the firm is currently producing its loss-minimizing quantity. If the firm represented...

Study smarter with the SolutionInn App


