An STM can also be operated in a mode in which electrons tunnel from the surface into
Question:
Figure 16.12
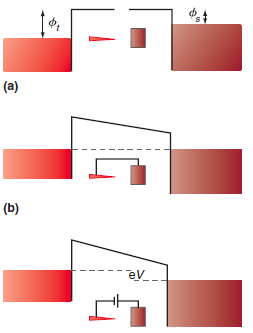
Transcribed Image Text:
(a) (b) ev
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
You would simply reverse the polarity of t...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A hole is drilled into the sphere of Problem 84 toward the center of the sphere to a depth of 2 km below the spheres surface. A small mass is dropped from the surface into the hole. Determine the...
-
a. What is the probability that an electron will tunnel through a 0.50 nm air gap from a metal to a STM probe if the work function is 4.0 eV? b. The probe passes over an atom that is 0.050 nm tall....
-
(a) For CH2O, run HF/3-21G geometry optimization and vibrational-frequency calculations to obtain the predicted geometry, dipole moment, and harmonic vibrational wavenumbers. Verify that all...
-
Water flows in a cylindrical, horizontal pipe. As the pipe narrows to half its initial diameter, the pressure in the pipe changes. (a) Is the pressure in the narrow region greater than, less than, or...
-
1. One of the events in a strength competition is to pull a cement block 100 feet. One competitor pulls the block by exerting a constant force of 250 pounds on a rope attached to the block at an...
-
Use the Gagnon 2016 income statement that follows and the balance sheet from SI3-6 to compute the following: a. Gagnon, Inc.'s, rate of inventory turnover and days' inventory outstanding for 2016. b....
-
Waring Corporation manufactures safeslarge mobile safes, and large walk-in sta- tionary bank safes. As part of its annual budgeting process, Waring is analyzing the profitability of its two products....
-
Why may an investor prefer a debenture with a put feature in preference to a bond with a call feature?
-
f(x)=x-12x+34x-16 The real zero(s) of f is/are 8,2+ 2,2-2 (Simplify your answer. Type an exact answer, using radicals as needed. Use integers or fractions for any numbers in the expression. Use a...
-
The following facts pertain to a non-cancellable lease agreement between Woodhouse Leasing Corporation and McKee Electronics Ltd., a lessee, for a computer system: The collectibility of the lease...
-
Explain how you can use size-quantized quantum dots to create a protein with a barcode that can be read using light.
-
For CdSe quantum dots, the emission wavelength increases from 450. nm to 650. nm as the dot diameter increases from 2 to 8 nm. Calculate the band gap energy for these two particle diameters.
-
True Or False Day-in-the-life videos are generally inadmissible because they unfairly appeal to jurors sympathies.
-
The Green Cab Company charges $2.50 plus $0.45 per quarter mile traveled. The Red Taxi Company charges $1.70 plus $0.55 per quarter mile. Apparently, you should take a Red Taxi for short trips and a...
-
Based on the module resources, think about the trends that are occurring in the healthcare marketplace and determine a new service line that your organization can add to increase revenues or save...
-
Pongo Sports Corporation sells two lines of table tennis paddles. Monthly sales and the contribution margin ratios are shown below: Slammo Pingy Total Company Sales Revenue $75,000 $225,000 30%...
-
You will chose a partner and research one of the drugs listed below(Haloperidol). You will state the following: What is the purpose of the drug? What are the side effects of the drug? What...
-
17. Which of the following is an active transport mechanism? a. Osmosis b. Facilitated diffusion c. Filtration d. Pinocytosis 18. Which of the following transport mechanisms transports water across...
-
Show how you would convert hex-1-ene to each of the following compounds. You may use any additional reagents and solvents you need. (a) 2-methoxyhexane (b) 1-methoxyhexane (c) 1-methoxyhexan-2-ol (d)...
-
The manager of a local convenience store is expanding his line of small toy items. To price these new items, the manager is looking at the prices being charged by competing retailers in his area. For...
-
Can molecules belonging to the point groups D 2h or C 3h be chiral? Explain your answer.
-
Identify the point groups to which all isomers of dichloronaphthalene belong.
-
What states of (i) benzene, (ii) naphthalene may be reached by electric dipole transitions from their (totally symmetrical) ground states?
-
Comprehensive Problem (Algo) LO 11-1, 11-2, 11-3, 11-4 Benson Modems, Inc. acquired a subsidiary named Anywhere, Inc. (Al). Al manufactures a wireless modem that enables users to access the Internet...
-
1The building was purchased on May 3, 2009, and is depreciated to the nearest whole month using the straight-line method. Depreciation is based on a 15-year life, after which it will be demolished...
-
With respect to the adjustments made by the analyst in Country B related to property, plant, and equipment, answer the following: 1. Why does the adjustment for depreciation related to revalued...

Study smarter with the SolutionInn App


