Explain the direction of the chemical shifts for Fe(0), Fe(II), and Fe(III) in Figure 22.20. Fe 2p
Question:
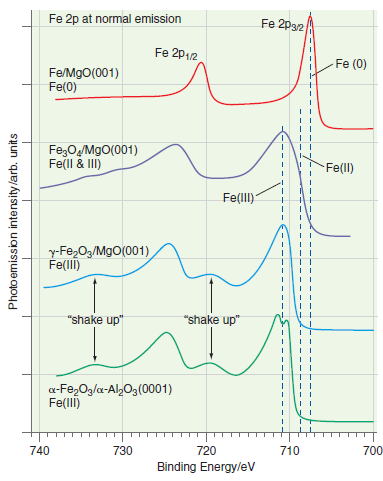
Transcribed Image Text:
Fe 2p at normal emission Fe 2p32 Fe 2p12 Fe (0) Fe/MgO(001) Fe(0) Fe;OM9O(001) Fe(ll & II) Fe(ll) Fe(lI) y-Fe,O3/MgO(001) Fe(llI) "shake up" "shake up" a-Fe,Og/a-Al2Og (0001) Fe(llI) 740 730 720 710 700 Binding Energy/ev Photo emission intensity /arb. units
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
As electrons are removed from Fe to form Fe 2 or Fe 3 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
The chemical shifts of the 1H nuclei in 2,2-dimethylpropane and TMS are 0.95 and 0.0, respectively. From these data, what can you deduce about the relative electronegativities of carbon and silicon?
-
A non reactive/conservative contaminant is dumped on the ground level and it leaches to the groundwater vertically and takes half day for reaching the groundwater by travelling through unsaturated...
-
The point is on the terminal side of an angle in standard position. Find the exact values of the six trigonometric functions of the angle. 1. (12, 16) 2. (7, -24) 3. (-0.5, 4.5)
-
Chen Company's Small Motor Division manufactures a number of small motors used in household and office appliances. The Household Division of Chen then assembles and packages such items as blenders...
-
Why is capital investment appraisal strategically important to an organisation?
-
Soto Industries Inc. is an athletic footware company that began operations on January 1, Year 1. The following transactions relate to debt investments acquired by Soto Industries Inc., which has a...
-
The equation below gives parametric equations and parameter intervals for the motion of a particle in the xy-plane. Identify the particle's path by finding a Cartesian equation for it. Graph the...
-
Plum Corporation began the month of May with $700,000 of current assets, a current ratio of 2.50:1, and an acid-test ratio of 1.10:1. During the month, it completed the following transactions (the...
-
The principal line in the emission spectrum of sodium is yellow. On close examination, the line is seen to be a doublet with wavelengths of 589.0 and 589.6 nm. Explain the source of this doublet.
-
Why is XPS a surface-sensitive technique?
-
How is a functions differentiability at a point related to its continuity there, if at all?
-
Determining the exact concentration of HC1. Based on the information below, what can you conclude about this lab? What is the deviation? Results: THAM molar mass: 121.1 g/mol Sample 1 Sample 2 Sample...
-
Karen-Cook Corporation issued 290,000 shares of $20 par value, 6% preferred stock on January 1, 2018, for $6,525,000. In December 2020, Karen-Cook declared its first dividend of $870,000. (a) Prepare...
-
Given the following expected returns and standard deviations: Expected Return Standard Deviation Asset A 15% Asset B 30% Calculate and plot the efficient frontier for each of the following...
-
The Miami MarlinsThe ProblemThe Miami Marlins made the groundbreaking decision in 2020 to hire KimNg to be the general manager of the team. She is believed to be recognized as thefirst female GM in...
-
The following T-accounts record the operations of Waterway Co.: Assume all raw materials are direct materials. Raw Materials Beginning Balance 11,112 ????? 66,998 Ending Balance 13,322 Work in...
-
Dharma, a public company, sold a piece of equipment at the beginning of Year 1, receiving a $10,000, two-year 2% note. Interest is paid at the end of each year. Market interest rates are assumed to...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
The heat capacity of chloroform (trichloromethane, CHCl 3 ) in the range 240 K to 330 K is given by C p,m /(J K 1 mol 1 ) = 91.47 + 7.5 10 2 (T/K). In a particular experiment, 1.00 mol CHCl 3 is...
-
A sample consisting of 1.0mol CaCO 3 (s) was heated to 800C, when it decomposed. The heating was carried out in a container fitted with a piston that was initially resting on the solid. Calculate the...
-
A chemical reaction takes place in a container of cross-sectional area 100 cm 2 . As a result of the reaction, a piston is pushed out through 10 cm against an external pressure of 1.0 atm. Calculate...
-
Wendell's Hot Sauce uses a process costing system to determine its product's cost. The last of the three processes is packaging. The Packaging Department reported the following information for the...
-
Consider the following data for two products of Gitano Manufacturing. (Round your intermediate calculations and OH rate and cost per unit answers to 2 decimal places) Number of units produced Direct...
-
Mr. Amru Nazli started the business of a gold shop named Emas Ria. The store started operating on 1st Jan 2021. Here are the transactions for the month first store operations. 1-Jan-21 Starting a...

Study smarter with the SolutionInn App


