From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data
Question:
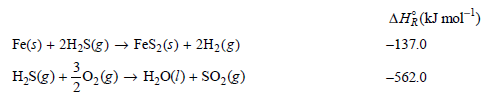
Transcribed Image Text:
AĦR(kJ mol) Fe(s) + 2H2S(g) –→ FeS2(s) + 2H2(g) -137.0 H;S(g) + 0,(g) → H,O() + SO,(E) -562.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
HOl SOg HSg 20 8 Ss Og SO g Hg Og HO1 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard enthalpy of formation for diamond, given that C(graphite) + O2(g) CO2(g) Afr =-393.5 kJ/mol C(diamond) + O2(g) CO2(g) AF1 395.4 kJ/mol
-
From the following data at 298.15 K calculate the standard enthalpy of formation of FeO(s) and of Fe 2 O 3 (s): A(kJ mol) Fe,0;(s) + 3C(graphite) 2Fe(s) + 3cO(g) FeO(s) + C(graphite) Fe(s) + CO(g)...
-
From the enthalpy of formation for CO2 and the following information, calculate the standard enthalpy of formation for carbon monoxide (CO). Why can't we obtain it directly by measuring the enthalpy...
-
The following items are dropped from an airplane. Rank them in order from lowest terminal speed to highest and justify your ranking. (a) Bowling ball (b) Beach ball (c) Spear or javelin (pointing...
-
San Antonio Company produces accounting software. Its unit cost structure, based on an anticipated production volume of 150,000 units, is:sale price is 160, variable costs is 60 and fixed costs is...
-
How much energy is required to ionize a hydrogen atom initially in the n = 2 state?
-
Use the method of eigenfunction expansions to solve the problems: a. \(y^{\prime \prime}+4 y=x^{2}, \quad y^{\prime}(0)=y^{\prime}(1)=0\). b. \(y+4 y=x^{2} \quad y(0)=y(1)=0\).
-
Sepracor, Inc., a U.S. drug company, reported the following information. The company prepares its financial statements in accordance with U.S. GAAP . 2007 (, 000) Current liabilities $ 554,114...
-
doall requirements Data tableRequirements 1. Recast the income statement to emphasize contribution margin. 2. Calculate the contribution margin percentage and breakeven point in units and revenues...
-
Harriet's Hats is a retailer who buys hats from a manufacturer and then sells them in its stores. The following information includes the company's December 31, 20Y8 Balance Sheet and the details of...
-
Which of Ne or Ar has the larger van der Waals parameter a? Explain your reasoning.
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
Describe how prototyping can be used to augment the traditional SDLC.
-
7.If you had done this lab on the moon, where g = 1.60 m/s 2, would your measured times be longer, shorter or about the same? Compute the time expected if m2 = 560 grams and m1 = 550 grams and Ay =...
-
Mirada Company manufactures handheld calculators and has the following information available for the month of July: Work in process, July 1 ( 1 0 0 % complete for materials, 2 5 % for conversion ) 1...
-
We have the following Cagan equation:
-
What is the purpose of financial markets? How can this purpose be accomplished efficiently? A./Discuss the functions of financial intermediaries. 15. A number of factors give rise to different...
-
Monthly loan payment Personal Finance Problem Tim Smith is shopping for a used luxury car. He has found one priced at $35,000. The dealer has told Tim that if he can come up with a down payment of...
-
Draw the graph of a one-to-one function that contains the points (-2, -3), (0, 0), and (1, 5). Now draw the graph of its inverse. Compare your graph to those of other students. Discuss any...
-
4. Jobe dy -Y 2 et by
-
A lot of information about the energy levels and wave functions of small inorganic molecules can be obtained from their ultraviolet spectra. An example of a spectrum with considerable vibrational...
-
Aromatic hydrocarbons and 12 form complexes from which charge transfer electronic transitions are observed the hydrocarbon acts as an electron donor and 12 as an electron acceptor the energies hvmax...
-
Consider some of the precautions that must be taken when conducting single-molecule spectroscopy experiments. (a) What is the molar concentration of a solution in which there is, on average, one...
-
In addition to calculating earnings, deductions and employer obligations arising from payroll, payroll administrators must ensure that all liabilities resulting from the payroll process are paid on...
-
2. Answer chapter 5 Study Questions and Problems in the ASP for question numbers 1, 2, 3, 4, 5,6,7,8,9,11,12,13,16 on page 83.
-
Potential output is the level of output when all _ _ _ _ _ _ _ _ are in equilibrium.

Study smarter with the SolutionInn App


