The gas-phase decomposition of ethyl bromide is a first-order reaction, occurring with a rate constant that demonstrates
Question:
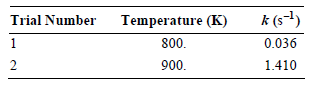
a. Determine the Arrhenius parameters for this reaction.
b. Using these parameters, determine ΔH€¡ and ΔS€¡ as described by the Eyring equation.
Transcribed Image Text:
k (s-l) Trial Number Temperature (K) 0.036 800. 900. 1.410
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a The activation energy is determined by taking the ratio of the rate constan...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The unimolecular decomposition of urea in aqueous solution is measured at two different temperatures and the following data are observed a. Determine the Arrhenius parameters for this reaction. b....
-
Hydrogen abstraction from hydrocarbons by atomic chlorine is a mechanism for Cl · loss in the stratosphere. Consider the reaction of Cl· with ethane: C 2 H 6 (g) + Cl· (g) C 2 H...
-
Chlorine monoxide (ClO i ) demonstrates three bimolecular self-reactions: The following table provides the Arrhenius parameters for this reaction: a. For which reaction is ÎH ¡ greatest...
-
answer the following questions about the countries below using CIA factbook Economic Systems Continuum Activity: Country Information Cards Answer the following questions about the countries below...
-
Find each function value. 1. f (x) = |x + 2| - 15, (a) f (-8) (b) f (14) (c) f (x - 6) 2. f (x) = (x + 9) / (x2 - 81) (a) f (7) (b) f (-5) (c) f (x - 9)
-
Members of the millennial generation are continuing to be dependent on their parents (either living with or otherwise receiving support from parents) into early adulthood (the Enquirer, March 16,...
-
Is a valuation of a cinema theatre or a chemist shop in terms of a number of weeks sales a result of the sum-of-the-parts or the cash flow method?
-
ABC Audio sells headphones and would like to earn after-tax profits of $400 every week. Each set of headphones incurs variable costs of $5 and sells for $10. Rent and other fixed costs are $200 per...
-
Access an online loan calculator with annual payments, such as the one at mycalculators.com, to produce an amortization schedule for Welton Corporations installment note that has original principal...
-
Base plc acquired 60% of the common shares of Ball plc on 1 January 20X0 and gained control. At that date the statements of financial position of the two companies were as follows: Note: The fair...
-
Catalase is an enzyme that promotes the conversion of hydrogen peroxide (H 2 O 2 ) into water and oxygen. The diffusion constant and radius for catalase are 6.0 10 7 cm 2 s 1 and 51.2 ,...
-
Consider the unimolecular isomerization of methylcyanide, a reaction that will be discussed in detail in Chapter 36: CH 3 NC(g) CH 3 CN(g) The Arrhenius parameters for this reaction are A = 2.5 10...
-
Students at Hunter College studied bicycle safety in New York City (Tuckel & Milczarski, 2014). They reported data on cyclists who were riding their own bikes and were not cycling as part of their...
-
In the first lecture you were told that "a company is an artificial legal person". Snide has a business as a shoe maker and conducts it in his own name. Then he registers with ASIC a company called...
-
Consider a no-load mutual fund with $252 million in assets, 20 million shares and $36 million in debt at the start of the year. It invests in a portfolio that provides no income but increases price...
-
Find the quadratic polynomial whose graph passes through the points (1, 12), (2, 29), (3, 54). y = NOTE: Use the variable x in your answer.
-
A small rock with mass 0.10 kg is released from rest at point. A, which is at the top edge of a large, hemispherical bowl with radius R=0.60 m (the figure (Figure 1)). Assume that the size of the...
-
You sell your house at 95.7% of the list price. What was the amount of sales commission if the commission percentage is 3.75% and the list price of the house was $182 500?
-
Sharpton, the engraving department is a bottleneck, and the company is considering hiring an extra worker, whose salary will be $65,000 per year, to mitigate the problem. With the extra worker, the...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
As shown in Example Problem 3.5, (U m /V) T a/V 2 m for a van der Waals gas. In this problem, you will compare the change in energy with temperature and volume for N 2 , treating it as a van der...
-
Calculate H o f for NO(g), at 975 K, assuming that the heat capacities of reactants and products are constant over the temperature interval at their values at 298.15 K.
-
Prove that C V = - (U/V) T (V/T) U.
-
Oxford Company has limited funds available for investment and must ration the funds among four competing projects. Selected information on the four projects follows: Life of Investment Present Value...
-
a. Assuming that the desired rate of return is 15%, determine the net present value for the proposal. Use the table of the present value of $1 presented above. If required, round to the nearest...
-
Delta Company produces a single product. The cost of producing and selling a single unit of this product at the company's normal activity level of 108,000 units per year is: Direct materials Direct...

Study smarter with the SolutionInn App


