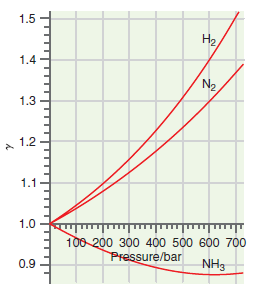
What can you conclude about the ratio of fugacity to pressure for N 2 ,H 2 ,
Question:
Figure 7.10
Transcribed Image Text:
1.5 H2, 1.4 N2 1.3 1.2 1.1 1.0 100 200 300 400 500 600 700 Pressure/bar 0.9 NH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
F P for ...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If A = B, what can you conclude about the components of A and B?
-
You are interested in discerning the role of Ï factors in prokaryotic transcription, using a system of purified core polymerase, a DNA template, a Ï factor, and labeled nucleotides...
-
What can you conclude about the interaction between ions in an electrolyte solution if the mean ionic activity coefficient is greater than one?
-
A person who is 6 feet and 3 inches tall and weighs 185 pound force (lbf) is driving a car at a speed of 65 miles per hour over a distance of 25 miles between two cities. The outside air temperature...
-
Shady Lane's Income tax payable account decreased from $14.3 million to $11.7 million during 2016. If its Income tax expense was $80.2 million, what was shown a an operating cash flow under the...
-
Two magnifying glasses are labeled with their angular magnifications. Glass A has a magnification of "2 " (M = 2) and glass B has a magnification of 4 . Which has the longer focal length? Explain.
-
7. Gilleo opposed American participation in the war in the Persian Gulf. She displayed a large sign on her front lawn that read, "Say No to War in the Persian Gulf, Call Congress Now." The city of...
-
The reported net incomes for the first 2 years of Sandra Gustafson Products, Inc., were as follows: 2014, $147,000; 2015, $185,000. Early in 2016, the following errors were discovered. 1....
-
??????? The most recent financial statements for Crosby, Incorporated, follow. Interest expense will remain constant; the tax rate and the dividend payout rate will also remain constant. Costs, other...
-
Case 1. Presented below are the financial balances for the BonGiovi Company and the Terens Company as of December 31, 2017, immediately before BonGiovi acquired Terens. Also included are the fair...
-
Predict the major product of the reaction between 1-butanol and: (a) PBr 3 (b) SOCl 2 , py (c) HCl, ZnCl 2 (d) H 2 SO 4 , heat (e) PCC, CH 2 Cl 2 (f ) Na 2 Cr 2 O 7 , H 2 SO 4 , H 2 O (g) Li (h) NaH...
-
A system consisting of 82.5 g of liquid water at 300. K is heated using an immersion heater at a constant pressure of 1.00 bar. If a current of 1.75 A passes through the 25.0 ohm resistor for 100. s,...
-
In Exercises determine which of the vectors is (are) parallel to z. Use a graphing utility to confirm your results. (a) (b) (c) (d) z = i j + k
-
A firm is considering a project that costs $165,000 to start and generates $28,000/year in after-tax cash for the next 8 years. After 8 years there are no additional cash flows or salvage value. The...
-
SecuriCorp operates a fleet of armored cars that make scheduled pickups and deliveries in the Los Angeles area. The company is implementing an activity-based costing system that has four activity...
-
The Houdini Magic Shoppe has forecast its sales and purchases for the last 4 months of 2020. Sales are as follows (September to December): 2100, 1300, 2300, 2800 Purchases are as follows (September...
-
Golden Wedding Dress Company designs custom wedding dresses for brides to be. The person preparing the adjusting entries at year-end was unable to complete the adjustments due to illness. You have...
-
the following information is available for ADT Company which produces Special order security products and uses a job order costing system. overhead is applied using a predetermined overhead rate of...
-
Make up an arithmetic sequence. Give it to a friend and ask for its twentieth term.
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
Which of the molecules in Exercises 12.9b and 12.10b can be? (a) Polar, (b) Chiral?
-
Consider the C3Yion NO;. Is there any orbital of the central N atom that can have a nonzero overlap with the combination 2pz (A) pz (B) pz (C) of the three O atoms (with z perpendicular to the...
-
The CIO2 molecule (which belongs to the group C, 2v) was trapped in a solid. Its ground state is known to be BJ Light polarized parallel to the y-axis (parallel to the 00 separation) excited the...
-
heoretically, the fed funds rate cannot be______ the discount rate or________ the rate paid on reserves. A. above, below B. below, above C. below, below D. above, above
-
Abacus... a.) is primarily a business bank b.) was and is located in the United States and China c.) was established to meet the depository and borrowing needs of local customers in a certain...
-
Efficient Portfolios are those which offer OA. Highest expected return for a given level of risk B. The minimum risk and expected return OC. The maximum expected return and risk OD. Highest risk for...

Study smarter with the SolutionInn App


