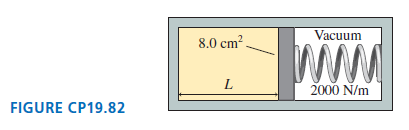
A monatomic gas fills the left end of the cylinder in Figure CP19.82. At 300 K, the
Question:
Transcribed Image Text:
8.0 cm?. Vacuum 2000 N/m FIGURE CP19.82
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Model Assume the gas is ideal Solve Apply the first law of thermodynamics Eth W Q nC V T Consider ad...View the full answer

Answered By

Joseph Njoroge
I am a professional tutor with more than six years of experience. I have helped thousands of students to achieve their academic goals. My primary objectives as a tutor is to ensure that students do not have problems while tackling their academic problems.
4.90+
10+ Reviews
27+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Helium (He), a monatomic gas, fills a 0.010-m3 container. The pressure of the gas is 6.2 105 Pa. How long would a 0.25-hp engine have to run (1 hp = 746 W) to produce an amount of energy equal to...
-
A monatomic gas and a diatomic gas both have n moles and are at temperature T. What is the difference in their internal energies? Express your answer in n, R, and T.
-
Repeat Problem 72 for a monatomic gas. B. D.
-
Generics, Inc. is a U. S. GAAP reporter that manufactures and sells generic drugs and has a December 31 year-end. On March 1, 2014, it began selling a drug, Anocyn, which is a generic of Dicital....
-
On August 10, 2014, Jasper purchased business equipment for $40,000. On his 2014 tax return, $40,000 of 179 immediate expense was taken on the equipment. On July 14, 2015, Jasper sold the equipment...
-
You have just been transferred from the Montreal office to the Vancouver office of your company, a national sales organization of electrical products for developers and contractors. In Montreal, team...
-
Win is it necessary to use comparative balance sheets, a current income statement, and certain transaction data in preparing a statement of cash flows.'
-
Valmont, Inc., experienced the following events in 2012, in its first year of operation. 1. Received $20,000 cash from the issue of common stock. 2. Performed services on account for $50,000. 3. Paid...
-
A couple wishes to borrow money using the equity in their home for collateral. A loan company will loan them up to 70% of their equity. They puchased their home 10 years ago for $65, 757. The home...
-
The totals line from Nix Company's payroll register for the week ended March 31, 20--, is as follows: Payroll taxes are imposed as follows: Social Security tax, 6.2%; Medicare tax, 1.45%; FUTA tax,...
-
0.020 mol of a diatomic gas, with initial temperature 20C, are compressed from 1500 cm 3 to 500 cm 3 in a process in which pV 2 = constant. How much heat energy is added during this process?
-
Solids and liquids resist being compressed. They are not totally incompressible, but it takes large forces to compress them even slightly. If it is true that matter consists of atoms, what can you...
-
It is May 5, 2014. The quoted price of a government bond with a \(12 \%\) coupon that matures on July 27, 2024, is \(110-17\). What is the cash price?
-
Carbon dioxide (CO2) fills a closed, rigid tank fitted with a paddle wheel, initially at 80\ deg F, 50 lbf/in 2, and a volume of 1.6 ft3. The gas is stirred until its temperature is 500\deg F. During...
-
Determine the four missing amounts using financial statement relationships. Revenues Expenses Net income Dividends declared during the year Retained earnings Beginning Ending Total assets Beginning...
-
1 Find the arc length for the curve defined by y=x23 over the x-interval [0,8] 2. Turn in: Clearly construct two integrals to find the arc length of y=-Inx for ye [0,2] as shown. One with respect to...
-
A drainage ditch alongside a highway with a 3% grade (drops 3 mfor every 100 m of length) has a rectangular cross section of depth1.35 m and width 2.5 m. To prevent soil erosion, the ditch isfilled...
-
Assess today's risk premium on industrial bonds using the Bonds Online Web site. (https://www.bondsonline.com/) Select the Today's Market section and then choose Corporate Bond Spreads . Use the...
-
Elaine runs the Five Brothers restaurant in Niagara Falls. She has given the bank a general security interest on the restaurant property that she owns in exchange for a loan of $200 000. The...
-
F.(3e* -2x 3 sin(2x)) is equal to 2 3 Cos 8. IT 3, t (4+@ 2 3, 1+o 1 4 Cos 4 4 1 3. 1 +4cos V7 (1+o 4 1 4 Cos 4 1+0 4-
-
What might the spectrum of an atom look like if the atoms electrons were not restricted to particular energy levels?
-
Some older cars vibrate loudly when driving at particular speeds. For example, at 65 mph the car may be most quiet, but at 60 mph the car may rattle uncomfortably. How is this analogous to the...
-
Place the proper number of electrons in each shell: Rubidium, Rb Sodium, Na Krypton, Kr Chlorine, CI
-
In developing a budget, which of the following should be considered? I. Balance sheet II. Chart of accounts III. Expenses from the current/past year IV. Purchasing contracts V. Physician compensation...
-
JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter 3Expected Sales Units150175Sales Price per Unit$250250$250otal Sales Revenue37,500$ JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter...
-
Which of the following statements is true concerning foreign income tax paid? Question 39Select one: a. If a taxpayer takes a subtraction on their Oregon return, then the subtraction is included as...

Study smarter with the SolutionInn App


