FIGURE P23.44 shows a thin rod of length L with total charge Q. Find an expression for
Question:
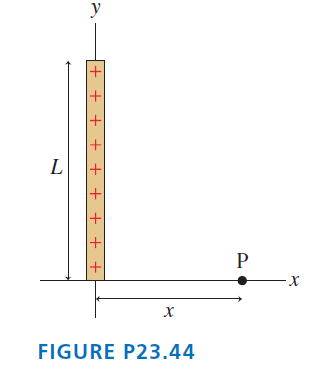
Transcribed Image Text:
P х FIGURE P23.44 + + + + + + + + +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Model The electric field is that of a line charge of length L Visualize Let the botto...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
FIGURE P25.69 shows a thin rod of length L and charge Q. Find an expression for the electric potential a distance z away from the center of rod on the line that bisects the rod. -Point on bisecting...
-
FIGURE P23.43 shows a thin rod of length L with total charge Q. a. Find an expression for the electric field strength at point P on the axis of the rod at distance r from the center.b. Verify that...
-
Figure shows a thin rod, of length L = 2.00 m and negligible mass, that can pivot about one end to rotate in a vertical circle. A ball of mass m = 5.00 kg is attached to the other end. The rod is...
-
Two different compounds have the formulation CoBr(SO4) 5NH3. Compound A is dark violet, and compound B is red-violet. When compound A is treated with AgNO3 (aq), no reaction occurs, whereas compound...
-
1. How successful do you think Ashley will be as a transformational leader at Ultra Cover? 2. What might Ashley be doing right as a transformational leader? 3. What suggestions might you offer Ashley...
-
The following spreadsheet shows the income statement of Bates Perfor 3 mance Auto: Requirement Write the appropriate formula in each cell that will need a formula. Choose from these symbols: + add -...
-
After closing its accounts at October 31,2010, Simmons Realty, Inc., had the following account balances: Prepare Simmons Realtys post-closing trial balance at October 31,2010. List accounts in proper...
-
Bottle-Up, Inc., was organized on January 8, 2010, and made its S election on January 24, 2010. The necessary consents to the election were filed in a timely manner. Its address is 1234 Hill Street,...
-
How would you use verbal and nonverbal concepts? Imagine you are preparing for a virtual job interview. How would a person make a professional persona for your interview and describe how you would...
-
At 37 C, the serine protease subtilisin has kcat = 50 s-1 and KM = 1.4 10-4 M. It is proposed that the N155 side chain contributes a hydrogen bond to the oxyanion hole of subtilisin. J. A. Wells and...
-
FIGURE P23.42 is a cross section of two infinite lines of charge that extend out of the page. The linear charge densities are ±Î». Find an expression for the electric field...
-
The article A Music Key Detection Method Based on Pitch Class Distribution Theory (J. Sun, H. Li, and L. Ma, International Journal of Knowledge-based and Intelligent Engineering Systems, 2011:165175)...
-
Laurel, Inc., and Hardy Corp. both have 7 percent coupon bonds outstanding, with semiannual interest payments, and both are priced at par value. The Laurel, Inc., bond has 2 years to maturity,...
-
What volume of water, in gallons would be required to fill a 750 mL container? 1 liter = 0.265 gallon Calculate the volume of millimeters for each of the following: 75.0 g of ammonia gas (density...
-
How do banks utilize robotic process automation (RPA) to streamline back-office operations, and what impact does it have on operational costs and customer satisfaction?
-
What is the role of core banking systems in modern banking operations, and how do they support seamless integration across various banking channels ?
-
How does the video define leadership, how does it define management? Where does leadership and management intersect and where do they vary? Do you believe leadership or management is more important?
-
Student's backpack has a mass of 9.6 kg. The student applies a force of 94.08 N [up] while walking through 1.4 km [E] to get to school. Calculate the work done by the student on the backpack
-
Graph each equation. (x) = x
-
Write an essay describing the differing approaches of nursing leaders and managers to issues in practice. To complete this assignment, do the following: 1. Select an issue from the following list:...
-
If an object has a smaller density than water, will the object stay fully submerged, partly submerged, or rise completely out of the water when it is released underwater? Explain.
-
A steady stream of water flowing in a narrow pipe reaches a point where the pipe widens. Does the speed of the water increase, decrease, or remain the same when the pipe widens? Explain.
-
Why does the stream of water flowing from a faucet often get more narrow as the water falls? Explain.
-
2. After scouting a number of locations, your financial analysts have settled on a desirable location for one of the firm's new manufacturing plants. However, the state income and local property...
-
The "one-time good deal" Cash-For-Clunkers program offered by the federal government proved a temporary boon for car dealers. In addition to this program, dealers were eager to add their own...
-
1. You are Chief Financial Officer for a company that sells contact lenses by mail order to a notional market. Despite the national presence, the firm's facilities are all located in one state. Your...

Study smarter with the SolutionInn App


