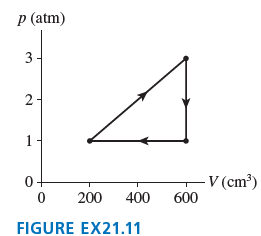
How much work is done per cycle by a gas following the pV trajectory of FIGURE EX21.11?
Question:
Transcribed Image Text:
p (atm) 3- 2- 1- V (cm³) 600 0+ 200 400 FIGURE EX21.11
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Model The work done by the gas per cycle ...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A gas following the pV trajectory of FIGURE EX21.12 does 60 J of work per cycle. What is p max ? P (kPa) Pmax 100 - V (cm) 0+ O 200 400 600 800 FIGURE EX21.12
-
(a) During each cycle, a Carnot engine absorbs 750 J as heat from a high-temperature reservoir at 360 K, with the low-temperature reservoir at 280 K. How much work is done per cycle? (b) The engine...
-
FIGURE P21.35 shows a heat engine going through one cycle. The gas is diatomic. The masses are such that when the pin is removed, in steps 3 and 6, the piston does not move. a. Draw the pV diagram...
-
1.The purpose of using a balanced scorecard is for: Multiple Choice A.companies to make sure their business activities align with strategy. B.companies to compare themselves with their competitors....
-
What are key considerations in monitoring deviations from performance standards?
-
Bauer and Do"pfmer (1999) examined the efficacy of lithium as an augmentation to conventional antidepressants. They found nine placebo-controlled studies testing lithium for those cases that did not...
-
Why would a defendant want to remove an action from state court to federal court? What is the procedure the defendant must follow in order to remove an action filed in state court?
-
Salamone Heaters selected data for October 2013 are presented here (in millions): Direct materials inventory, 10/ 1/ 2013 $ 75 Direct materials purchased 335 Direct materials used 380 Total...
-
3. [Maximum mark: 6] A company is designing a new logo. The logo is created by removing two equal segments from a rectangle, as shown in the following diagram. diagram not to scale The rectangle...
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
The first and second laws of thermodynamics are sometimes stated as You cant win and You cant even break even. Do these sayings accurately characterize the laws of thermodynamics as applied to heat...
-
What are (a) W out and Q H (b) The thermal efficiency for the heat engine shown in FIGURE EX21.13? p (kPa) 400 Q = -90 J 200 Q =-25 J V (cm) 0- 100 200 FIGURE EX21.13
-
Use the figure below, which shows the linear demand and constant cost conditions facing a firm with a high barrier to entry, to answer the following questions: a. The profit-maximizing price is $...
-
complete the following problems Do not rename provided data files or edit them in any way. Do not use global paths in your script. Instead, use setwd ( ) interactively in the console, but do not...
-
Determine the value of h such that the following system has infinitely many solutions. Solution: h = -48x + + 40 y = -24 42x + hy = 21
-
The following industries are to be used: hospitality & tourism, professional services, retail, financial services, or transportation. To Understand the interconnection between the organization's...
-
5q Solve q + 5 = 9 by completing the square. 3 (Use a comma to separate multiple solutions.) Provide your answer below: 9
-
Solve the exponential equation algebraically. Round your result to three decimal places. Use a graphing utility to verify your answer. 6(10-9-x) + 4 = 2401 x =
-
What does the Big Rip scenario assume about dark energy?
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
(a) Calculate the full-power bandwidth of the circuit of Fig. 9.59. (b) If this circuit is connected in a non-inverting unity-gain feedback loop, sketch the output waveform V o if V i is a sinusoid...
-
Repeat Problem 9.21 except use the aspect ratios, supply voltages, and bias current given in Fig. 6.58 instead of the values in Fig. 9.60. Also, assume that X d = 0.1 µm for all transistors...
-
If the circuit of Fig. 9.61 is used to generate the voltage to be applied to the gate of M 9 in Fig. 9.60, calculate the W/L of M 9 required to move the right half-plane zero to infinity. Use data...
-
Cash Supplies Land Building Total assets TYPHOON, INCORPORATED Balance Sheet December 31, 2024 Assets Liabilities $ 1,100 Accounts payable $ 3,400 Stockholders' Equity 5,400 Common stock 15,400...
-
DeSoto Tools Incorporated is planning to expand production. The expansion will cost $2,600,000, which can be financed either by bonds at an interest rate of 7 percent or by selling 52,000 shares of...
-
Waterway Inc. uses LIFO inventory costing. At January 1, 2025, inventory was $216,014 at both cost and market value. At December 31, 2025, the inventory was $283,252 at cost and $262,660 at market...

Study smarter with the SolutionInn App


