A solid mixture weighing 0.05485 g contained only ferrous ammonium sulfate and ferrous chloride. The sample was
Question:
A solid mixture weighing 0.05485 g contained only ferrous ammonium sulfate and ferrous chloride. The sample was dissolved in 1 M H2SO4, and the Fe2+ required 13.39 mL of 0.01234 M Ce4+ for complete oxidation to Fe3+ (Ce4+ + Fe2+ → Ce3+ + Fe3+).
Calculate the weight percent of Cl in the original sample.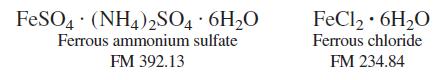
Transcribed Image Text:
FeSO4 · (NH4)2SO4 6H20 FeCl2 · 6H,O Ferrous ammonium sulfate Ferrous chloride FM 392.13 FM 234.84
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)

Answered By

Devendra Sharma
I have 22 years of teaching experience of chemistry for competitive exams such as NEET,IIT JEE. Also i have a deep teaching experience for different types of boards such as CBSE, ICSE.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A solid mixture weighing 0.5485 g contained only ferrous ammonium sulfate hexahydrate and ferrous chloride hexahydrate. The sample was dissolved in 1M H2SO4, oxidized to Fe3+ with H2O2, and...
-
A mixture contained aluminum sulfate and sodium sulfate. A sample of the mixture weighing 3.458 g was dissolved in water and treated with sodium hydroxide solution to yield a precipitate. The...
-
A mixture weighing 7.290 mg contained only cyclohexane, C6H12(FM 84.159), and oxirane, C2H4O (FM 44.053). When the mixture was analyzed by combustion analysis, 21.999 mg of CO2 (FM 44.010) were...
-
A process stream is heated using a shell and tube heat exchanger. The exit temperature is controlled by adjusting the steam control valve shown in figure. During an open-loop experimental test, the...
-
Based on data from the GSS10SDSS, we examine the relationship between highest educational degree and agreement to the statement, "Financial dependence on others is one of my greatest fears about old...
-
The Sweet Smell Fertilizer Company markets bags of manure labeled "not less than 60 lb dry weight." The packaged manure is a combination of compost and sewage wastes. To provide good-quality...
-
A business started its year owing 4,000 for electricity. During the year it paid electricity bills totalling 24,000 but owed 5,000 by the end of the period. What will be the figure transferred to the...
-
Coppola, who collected coins, joined a coin club, First Coinvestors, Inc. The club would send coins to its members, who were to pay for them or return them within 10 days. What was the nature of the...
-
The Bergie Restaurant Group manufactures the bags of frozen French fries used at its franchised restaurants. Last week, Bergie's purchased and used 103,000 pounds of potatoes at a price of $0.75 per...
-
Watershed is a media services company that provides online streaming movie and television content. As a result of the competitive market of streaming service providers, Watershed is interested in...
-
Consider the titration of 50.00 (0.05) mL of a mixture of I and SCN with 0.068 3 (0.000 1) M Ag. The first equivalence point is observed at 12.6 (0.4) mL, and the second occurs at 27.7 (0.3) mL. (a)...
-
Managing a salt-water aquarium. A tank at the New Jersey State Aquarium has a volume of 2.9 million liters. Bacteria are used to remove nitrate that would otherwise build up to toxic levels. Aquarium...
-
For what values of ' \(a\) ' does the system shown in figure has a zero steady state error [i.e., \(\operatorname{Lim}_{t ightarrow \infty} e(t)\) ] for a step input: (a) \(a=0\) (b) \(a=1\) (c)...
-
Define Procedural Fairness? When would procedural fairness arise? When would mediation be suitable? What is Proof of service? How is it used? What are the forms? How would a statute or regulation...
-
The home office ships merchandise to the branch at 45 percent above cost. If the balance before closing in the Intracompany Inventory Profit account is 76,000 and Shipments from Home Office amounted...
-
Q1. What are some measurable benefits from private equity ownership of corporations? Q2. What were the World Economic Forum's principal conclusions regarding private equity firms? Q3. What were the...
-
Luann Lawyer began working at 8:00A this morning. She called her client, Maizie Ruth Hill. They spoke for twenty (20) minutes about Maizie's case against Mike O'Dell. After the call ended, Luann...
-
When do you assess the quality of perishable stock on hand, such as chicken thighs and lettuce mix? Explain why you have chosen this timeframe.
-
Longboards Inc. was founded by Riley Thomas to produce a longboard he had designed for cruising. Longboards are catching up to skateboards in popularity because of their speed and durability. Up to...
-
Activator rod AB exerts on crank BCD a force P directed along line AB. Knowing that P must have a 100-N component perpendicular to arm BC of the crank, determine (a) The magnitude of the force P, (b)...
-
Explain how a galvanic cell uses a spontaneous chemical reaction to generate electricity.
-
Write a line notation and two reduction half-reactions for each cell pictured above. Fe Ag Pb Pb KOH(aq) K2SO4 (aq) H,S04(aq) FeO(s) Ag 0(s) PbSO4(s) PbSO,(s) PbO2(s) Galvanic cells for Problem 13.8
-
Draw a picture of the following cell and write reduction half- reactions for each electrode: Pt(s) | Fe*(aq), Fe*(aq) || Cr,O3 (aq), Cr*(aq), HA(aq) | Pt(s)
-
ABC Corporation acquires a new piece of equipment for $50,000 and uses it in ABC's manufacturing operations. A few months after ABC places the machine in service, it discovers that the equipment is...
-
Marbry Corporation has provided the following information concerning a capital budgeting project: After-tax discount rate 9% Tax rate 30% Expected life of the project 4 Investment required in...
-
5. On September 1, 2023, Oriole Corportation received 58800 cash from a tenant for one year's rent in advance and recorded the transaction with a credit to rent Revenue. The December 31, 2023,...

Study smarter with the SolutionInn App


