A solid sample weighing 0.237 6 g contained only malonic acid and aniline hydrochloride. It required 34.02
Question:
A solid sample weighing 0.237 6 g contained only malonic acid and aniline hydrochloride. It required 34.02 mL of 0.087 71 M NaOH to neutralize the sample. Find the weight percent of each component in the solid mixture. The reactions are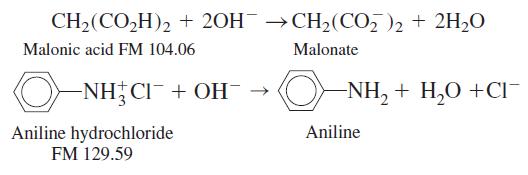
Transcribed Image Text:
CH2(CO,H)2 + 20H →CH2(CO, )2 + 2H,0 Malonic acid FM 104.06 Malonate -NH CI¯+ OH¯ → O- NH, + H,0 +CI¯ Aniline Aniline hydrochloride FM 129.59
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
3402 mL 08771 M NaOH 34020087711000 29810 3 mole of NaOH ...View the full answer

Answered By

TIRTHA MONDAL
I have completed my graduation and post graduation from Jadavpur University. I have also done Bachelor in Education (B.Ed) from the same university. I have experience in teaching JEE& NEET aspirants.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A solid mixture weighing 0.5485 g contained only ferrous ammonium sulfate hexahydrate and ferrous chloride hexahydrate. The sample was dissolved in 1M H2SO4, oxidized to Fe3+ with H2O2, and...
-
A mixture weighing 7.290 mg contained only cyclohexane, C6H12(FM 84.159), and oxirane, C2H4O (FM 44.053). When the mixture was analyzed by combustion analysis, 21.999 mg of CO2 (FM 44.010) were...
-
It takes 97.62 mL of 0.0546 M NaOH to titrate a 25.00 mL sample of H2SO4. What is the concentration of H2SO4? You will need to write the balanced chemical equation first.
-
The production of paper involves a pulping step to break down wood chips into cellulose and lignin. In the Kraft process, an aqueous, pulping-feed solution, known as white liquor, is used that...
-
Use the GSS 1OSSDS data file to study the relationship between the number of siblings a respondent has (SIBS) and his or her number of children (CHILDS). a. Construct a scatterplot of these two...
-
Jeff Kaufmanns machine shop sells a variety of machines for job shops. A customer wants to purchase a model XPO2 drilling machine from Jeffs store. The model XPO2 sells for $180,000, but Jeff is out...
-
A company -has opening inventory 6,900, closing inventory 7,800, purchases 33,650, carriage inwards 700 and purchases returns 400. What is its cost of sales? a. 31,650 b. 33,850 c. 33,050 d. 48,650
-
Weinberg Canning produces fillet, smoked salmon, and salmon remnants in a single process. The same amount of disposal cost is incurred whether a product is sold at split-off or after further...
-
he following information has been gathered for a company wanting to adopt the EOQ method: Annual demand in units 260.000 units Working days per year 260 days Lead time in days 12 Ordering cost per...
-
Using ratios to conduct financial analysis must be done with care. One year's worth of data is worth little; establishing a trend line over a period of years is the most useful approach. Using ratio...
-
A solution of NaOH was standardized by gravimetric titration of a known quantity of the primary standard, potassium hydrogen phthalate: Potassium hydrogen phthalate C 8 H 5 O 4 K, FM 204.22 The NaOH...
-
A 50.0-mL sample of 0.080 0 M KSCN is titrated with 0.040 0 M Cu + . The solubility product of CuSCN is 4.8 10 -15 . At each of the following volumes of titrant, calculate pCu + , and construct a...
-
Evaluate the following integrals. sec 2 d
-
9 assignment 03:32 ok Saved Help Save & Exit Check my w You have FIVE MINUTES remaining to complete your work On January 2, 2021, Archer Company, a skateboard manufacturer, installed a computerized...
-
Walt was a tax preparer who neglected to complete Form 8867 or record information needed for the return Rick fraudulently claimed the children for EIC purposes. After the IRS proved Rick had...
-
If an investment earns an interest rate of 3 % per year, compounded annually, and is worth $ 1 0 0 in 1 9 8 2 , what would it be worth in 2 0 2 2 ? ( Enter an answer correct to the nearest cent. ) .
-
A restaurant wants to expands its dining capacity. The building they want to acquire cost P400,000 and estimated a renovation cost of P180,000. The building will have an estimated salvage value of...
-
4. Suppose that f(x) = (x) using constants c and , and g(x) = O(x) using constants c2 and 2. We can prove that f(x)/g(x) = (x) using constants c3 and 3. Assume that g(x) is always strictly positive....
-
Financial data from the December 31 year-end statements of Sunrise Fashions are given below: Total assets . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
The voltage for the following cell is 0.490 V. Find Kb for the organic base RNH2. Pt(s) H2(1.00 bar) RNH2(aq, 0.10 M), RNH+3 Cl(aq, 0.050 M) ||' S.H.E.
-
The voltage of the cell shown here is - 0.246 V. The right half-cell contains the metal ion, M2+, whose standard reduction potential is - 0.266 V. Mc2+ + 2e - E° = - 0.266 V Calculate Kf for the...
-
The following cell was constructed to find the difference in Ksp between two naturally occurring forms of CaCO3(s), called calcite and aragonite.21 buffer(pH 7.00) CaCO3(s, aragonite) PbCO3(s) ...
-
If Total Liabilities increase, which of the following MUST be true?Group of answer choicesAssets MUST increaseAssets minus owners equity Must increaseOwners equity MUST increaseOwners equity minus...
-
I want write Introduction for my accounting assignment. I have chosen Pearson Corporation as my company and the same Introduction should be from accounting point of view. Also need to give...
-
Why does seasonality of sales pose particular problems for companies preparing interim reports? Select answer from the options below Annual sales cannot be reliably projected, causing variability in...

Study smarter with the SolutionInn App


