A solution was prepared from 10.0 mL of 0.100 M cacodylic acid and 10.0 mL of 0.080
Question:
A solution was prepared from 10.0 mL of 0.100 M cacodylic acid and 10.0 mL of 0.080 0 M NaOH. To this mixture was added 1.00 mL of 1.27 × 1026 M morphine. Calling morphine B, calculate the fraction of morphine present in the form BH1.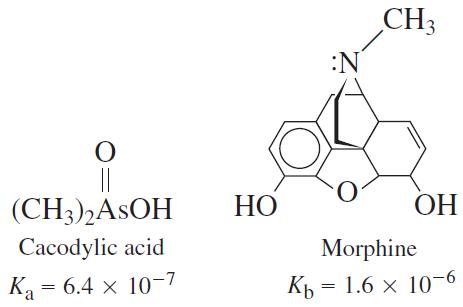
Transcribed Image Text:
CH3 :N || (CH3),ASOH НО ОН Cacodylic acid Morphine Ka = 6.4 x 10-7 Ка Къ — 1.6 х 10-6 Kp ||
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A solution was prepared by mixing 10.00 mL of unknown (X) with 5.00 mL of standard (S) containing 8.24 g S/mL and diluting the mixture to 50.0 mL. The measured signal quotient was (signal due to...
-
A solution was prepared from 1.023 g of the primary standard tris (Table 10 - 4) plus 99.367 g of water; 4.963 g of the solution were titrated with 5.262 g of aqueous HNO3 to reach the methyl red end...
-
A solution was prepared from 1.023 g of the primary standard tris (Table 10 - 4) plus 99.367 g of water; 4.963 g of the solution were titrated with 5.262g of aqueous HNO3 to reach the methyl red end...
-
Chuck, a single taxpayer, earns $86,000 in taxable income and $20,000 in interest from an investment in City of Heflin bonds. (Use the U.S. tax rate schedule.) Required: a. If Chuck earns an...
-
Use GSS 2010 to examine the relationship between respondents health (HEALTH) and social class (CLASS). Treat social class as the independent variable. a. Request the appropriate measures of...
-
Create a use case and an interaction diagram for scenarios that appeared in the above question.
-
Water originally saturated with carbon tetrachloride \(\left(\mathrm{CCl}_{4} ight)\) at \(25.0^{\circ} \mathrm{C}\) and \(1.0 \mathrm{~atm}\) is stripped with pure air at \(25.0^{\circ} \mathrm{C}\)...
-
Consider the following transactions of Pearl Software: Mar 31 Recorded cash sales of $180,000, plus sales tax of 8% collected for the state of Texas. Apr 6 Sent March sales tax to the state....
-
1.Oakleigh Corporation issued a corporate bond at par value of $1000. The current market price of the bond is $1120 and the term to maturity is 10 years.The current market yield on the corporate...
-
Design a CMOS op amp based on the folded-cascode architecture of Fig. 6.28 using supply voltages of ± 1.5 V. Use the bias circuit of Fig. 4.42 (with M 3 and M 4 cascoded) to generate the bias...
-
Calculate H2A , HA- , and A 2- for cis-butenedioic acid at pH 1.00, 1.92, 6.00, 6.27, and 10.00.
-
Explain what is wrong with the following statement: At its isoelectric point, the charge on all molecules of a particular protein is 0.
-
Determine the radius of gyration k y of the parabolic area. 160 mm y -y=0.1(1600-x) 40 mm X
-
Determine how much needs to be invested if you want to end up with $5000 in interest after 7 years of simple interest at 1.8% APR. Starting investment= 39682.54 dollars. Your answer must be accurate...
-
1. Write a VHDL Program for 1x4 Demultiplexer using two 1x2 Demultiplexer shown below using structural modelling. I 1x2 S 1x2 1x2 .Yo Y So Select Input Input S(1) S(0) F 00 (0) 01 y(1) 10 (2) 11 (3)...
-
What is the "Consumer Behavior and Marketing Mix" and why is it so important in strategy formulation and implementation?
-
What was Iris Inc.'s earnings before interest and taxes (EBIT)? Cost of good 320 Depreciation expense 35 Interest Expense 20 Operating Expense (excluding depreciation) 115 Sales 800
-
It is March 2017, and you were recently hired as a financial analyst at the private equity group Silver Capital Partners. Your boss tells you that he has heard that the management of Panera Bread, a...
-
a. Obtain a relative-frequency distribution for the classification (class-level) data. b. Using your answer from part (a), determine the probability that a randomly selected UWEC undergraduate...
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Consider the titration of 100.0 mL of 0.010 0 M Ce 4+ in 1 M HClO 4 by 0.040 0 M Cu+ to give Ce 3+ and Cu 2+ , using Pt and saturated Ag | AgCl electrodes to find the end point. (a) Write a balanced...
-
Calcium fluorapatite (Ca 10 (PO 4 ) 6 F 2 , FM 1 008.6) laser crystals were doped with chromium to improve their efficiency. It was suspected that the chromium could be in the +4 oxidation state. 1....
-
Primary-standard-grade arsenic(III) oxide (As 4 O 6 ) is a useful (but carcinogenic) reagent for standardizing oxidants including MnO 4 - and I-3. To standardize MnO 4 - , As 4 O 6 is dissolved in...
-
8 . Given the following FX market information, calculate the annualized 3 0 day, 9 0 day, and 1 8 0 day forward discounts for the Canadian dollar. Spot: C$ 1 = $ 1 . 0 0 7 8 3 0 day forward: C$ 1 = $...
-
Consider the two mutually exclusive projects: TimeAB 0(4500)(4600) 125902630 220102025 314651600 Calculate and draw out and label the 5 points on the NPV profile of the two projects.(10 points)Which...
-
Capital Budgeting (20 points) Samson is considering an investment of $500,000 in a machine with a five-year life. The machine will be depreciated straight-line to zero over five years. The machine...

Study smarter with the SolutionInn App


