Color Plate 4 shows how the color of the acid-base indicator bromocresol green (H 2 BG) changes
Question:
Color Plate 4 shows how the color of the acid-base indicator bromocresol green (H2BG) changes as NaCl is added to an aqueous solution of (H+)(HBG-). Explain why the color changes from pale green to pale blue as NaCl is added.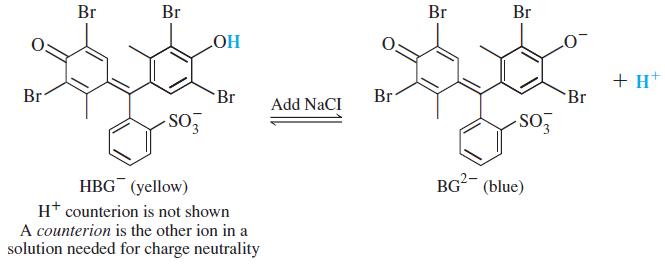
Transcribed Image Text:
Br Br Br Br Но + H* Br Br Br Br Add NaCI -SO BG (blue) HBG (yellow) H* counterion is not shown A counterion is the other ion in a solution needed for charge neutrality
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
On addition of NaCl the ionisabl...View the full answer

Answered By

Jyoti Chahal
I have done my graduation in Science from maharshi dayanand University, rohtak. I got 64.6% marks in bsc and masters in chemistry from Maharshi dayanand University with 64.5% marks. After that I did bachelor of education in science with passing marks of 69.4% from same University. I have given home tution also . Currently I am a subject matter expert on Transtutor.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The table in Figure 2.1 shows how the terms of the growth function for our dishwashing example are related to one another as n grows. Write a program that will create such a table for any given...
-
Heat is added to an ideal gas maintained at constant volume. Is it possible for the temperature of the gas to remain constant in this process? Explain.
-
Heat is added to an object initially at 30C, increasing its temperature to 80C. a. What is the temperature change of the object in Fahrenheit degrees? b. What is the temperature change of the object...
-
The maker of a $10,000, 8%, 90-day note receivable failed to pay the note on the due date of June 30. What accounts should be debited and credited by the payee to record the dishonored note...
-
Examine attitudes toward affirmative action based on two variables: AFFRMACT and DISCAFF. AFFRMACT measures respondents' support of preferential hiring and promotion of blacks (a higher score...
-
Kenneth and Patricia Golding spent a career as a husband-and-wife real estate investment partnership in Washington, DC. When they finally retired to a 25-acre farm in northern Virginias Fairfax...
-
During a month a business spends 195 on petty cash items and pays into petty cash a 5 note which the owner had borrowed previously. There was a cash float of 200 at the start of the month. Using the...
-
Charles Rubin is a 30-year employee of General Motors. Charles was pleased with recent negotiations between his employer and the United Auto Workers. Among other favorable provisions of the new...
-
View Policies Current Attempt in Progress Crane Timur is an accounting major at a midwestern state university located approximately 60 miles from a major city. Many of the students attending the...
-
Map the BANK ER schema of Exercise 3.23 (shown in Figure 3.21) into a relational schema. Specify all primary keys and foreign keys. Repeat for the AIRLINE schema (Figure 3.20) of Exercise 3.19 and...
-
Use Equation 7-12 to reproduce the curves in Figure 7-3. Plot your results on a single graph. F(Kp = 8.3 x 10-17) %3D 14 12 Br (Kp = 5.0 x 10-13) %D 10 CF (K = 1.8 x 10-10) sp -Br 2 10 20 30 40 50 60...
-
A 40.0-mL solution of 0.040 0 M Hg 2 (NO 3 ) 2 was titrated with 60.0 mL of 0.100 M KI to precipitate Hg 2 I 2 (K sp = 4.6 10 -29 ). (a) Show that 32.0 mL of KI are needed to reach the equivalence...
-
Compare the physical and chemical properties of H2O and H2S.
-
When It Comes to Eating Away at Democracy, Trump Is a Winner Links to an external site. . Thomas B. Edsall. The New York Times. 8/24/22 (segments of article) 1) After reading the article, how would...
-
Concentration of Carbon Monoxide (CO) in the Air According to a joint study conducted by Oxnard's Environmental Management Department and a state government agency, the concentration of CO in the air...
-
-At 0 ing i A wave on a string is described by y (x, t) = (2.0 cm) x cos [2 (x/(2.4 m) + t/(0.30 s))], where x is in m and t is in s. Part D What is the wave length? Express your answer in meters. =...
-
Eli has demonstrated an impressive ability to adjust to her new surroundings and has shown increasing interest in her environment. Her interactions with his peers and educator, her eager...
-
A stock price at time t is $13.95 and at time t +1is $15.50. What is the simple return for this time period at time t +1?Write the return as a decimal, not a percentage and round to two decimal...
-
Armcor Inc. is in a rapidly growing technological industry. Would you expect the company to have a high or low dividend payout ratio?
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
Why do glass pH electrodes tend to indicate a pH lower than the actual pH in strongly basic solution?
-
Suppose that the Ag | AgCl outer electrode in Figure 14-11 is filled with 0.1 M NaCl instead of saturated KCl. Suppose that the electrode is calibrated in a dilute buffer containing 0.1 M KCl at pH...
-
(a) When the difference in pH across the membrane of a glass electrode at 25C is 4.63 pH units, how much voltage is generated by the pH gradient? (b) What would the voltage be for the same pH...
-
Angela is entitled to a fixed annual payment of $15,000 from ABC Partnership without regard to the income of the partnership. Her share of the partnership income is 10%. After deducting the...
-
Rosie has $24,000 short-term capital loss, $10,500 28% gain and $9,000 0%/15%/20% gain. Which of the following statements is correct? a. Rosie has a $19,500 net capital gain b. Rosie has a $7,500...
-
Wyatt Company was formed on January 1, Year 1, when it acquired $50,000 cash from issuing common stock. Which of the following shows the impact of this transaction on Wyatt's accounting equation?...

Study smarter with the SolutionInn App


