Dry primary-standard-grade potassium hydrogen phthalate for 1 h at 110 o C and store it in a
Question:
Dry primary-standard-grade potassium hydrogen phthalate for 1 h at 110oC and store it in a desiccator.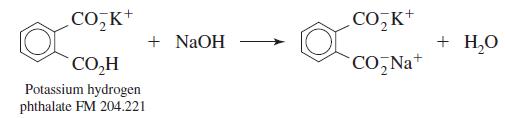
Transcribed Image Text:
CO,K+ CO, K+ + NaOH + H,0 CO,H CO, Na+ Potassium hydrogen phthalate FM 204.221
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

Akash Bongane
“I like to encourage students to ask questions and really understand subjects. I find non-traditional methods, like hands-on learning, to be particularly effective.”
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Buoyancy correction factor: mtrue/mread = 1.000 3244] [Interpolated density of water at 23.3 C: 0.997 468 9 g/mL] [HCl concentration at 20 C: 0.102343 M] Reagents: The molar mass and density of...
-
Potassium hydrogen phthalate is a primary standard used to measure the concentration of NaOH solutions. Find the true mass of potassium hydrogen phthalate (density = 1.636 g/mL) if the mass weighed...
-
Potassium hydrogen phthalate (KHP) is often used as a primary standard in acid-base titration. If 19.15 mL of NaOH is required to neutralize 0.442 g of KHP, what is the concentration of the NaOH?
-
Write a film script using this title "The arrival". THE ARRIVAL' WRITE A FILM SCRIPT USING THIS, TITLE DUMBFOUND: ASTONISH, AMAZE E62-67 SIMILE A METAPHOR ALLITERATION LIST OF THREE A LINE FROM A...
-
Segregation in schools appears to be increasing, due in part to class (and racial) separation between public and private schools. The table below summarizes the percentage of students who are white...
-
The set vice times for a new data entry clerk have been measured and sequentially recorded as shown below: Report Time (minutes) 1 ............ 66 2 ............ 56 3 ............ 53 4 ...............
-
Bioldley Brothers sell luxury picnic hampers from their prestigious shop in London. When the firm 's accountant drew up the list of receivables ledger balances at the business's year-end, 31 May...
-
A partially completed worksheet for Christopher Cobb, CPA, for the month ending June 30, 2016, is shown below. INSTRUCTIONS 1. Record the balances as of June 30 in the ledger accounts. 2. Prepare the...
-
Tesla, Inc. manufactures electric vehicles using mixed costing. In May 2034, the company incurs the following costs related to its production and distribution: Direct Materials: $600 million Direct...
-
1. Does this grant present Dr. Ray with an ethical dilemma(s) in any way? 2. Derive at least one hypothesis for each research question listed above. Provide a sound rationale or theoretical...
-
The inside cover of this book tells us that 8.2 mL of 37 wt% HCl should be added to 1 L of water to produce 0.1 M HCl. Prepare this solution in a capped polyethylene bottle, using a graduated...
-
Boil 1 L of water for 5 min to expel CO 2 . Pour the water into a polyethylene bottle, which should be tightly capped whenever possible. Calculate the volume of 50 wt% NaOH needed (5.3 mL) to produce...
-
Should the tax dollars of citizens of democratic nations be used to help people in authoritarian nations get around the Web censorship of their repressive governments?
-
Teams are a vital force in today's workplace. Although a few companies avoid team-based models, most companies consider teamwork fundamental to their success. Organizations form teams because...
-
A firm issued a $5,000 par value, 10-year maturity bond 2 years ago. The bond currently sells for $5,550.31 and has an 8 percent annual coupon. The firm's tax rate is 33 percent. The firm's after-tax...
-
2.You are considering buying shares in a growth stock that you believe will earn $2 per share next year, $4 in the following year and $6 in the third year (with all earnings paid out as...
-
Research Intergrated Marketing Communications (IMC) and discuss whether this strategy can be implemented in a small business such as a local restaurant. Your textbook provides examples as to how...
-
Do strategic business units often have their own missions and objectives?Explain
-
What is a perpetual budget?
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
Iodometric analysis of high-temperature superconductor. The procedure in Box 15-3 was carried out to find the effective copper oxidation state, and therefore the number of oxygen atoms, in the...
-
Here is a description of an analytical procedure for superconductors containing unknown quantities of Cu(I), Cu(II), Cu(III), and peroxide (O 2 2- ): 32 "The possible trivalent copper and/or...
-
Li 1 + y CoO 2 is an anode for lithium batteries. Cobalt is present as a mixture of Co(III) and Co(II). Most preparations also contain inert lithium salts and moisture. To find the stoichiometry, Co...
-
[STAT6032 ONLY] Use the Bonferroni method to compare the following treatments (i.e. these four specific level combinations of speed and depth): (2,3), (2,4), (3,3) 6%). Calculate the critical...
-
Provide a purchase journal entry of a product (50$ above), which is comprised of a rational cognition purchase decision, extensive problem-solving decision, ideal state opportunity recognition,...
-
12345 1 Income Statement 2021 2020 2019 2018 Period Ending: 30/06 30/06 30/06 30/06 5 Total Revenue 671.74 598.85 784.51 777.95 6 Revenue 671.74 598.85 784.51 777.95 7 Other Revenue, Total 8 Cost of...

Study smarter with the SolutionInn App


