Explain why each voltammogram from the rotating disk electrode in Figure 17-16 reaches a plateau at low
Question:
Explain why each voltammogram from the rotating disk electrode in Figure 17-16 reaches a plateau at low potential and at high potential. What chemistry occurs in each plateau? Why do all the curves overlap at low potential? How might the current density in each plateau change if the rotation speed were decreased?
In Figure 17-16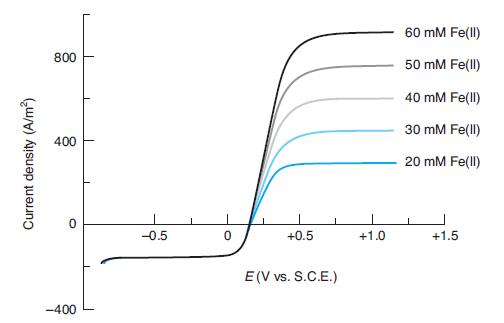
Transcribed Image Text:
60 mM Fe(ll) 800 50 mM Fe(ll) 40 mM Fe(ll) 30 mM Fe(ll) 400 20 mM Fe(ll) -0.5 +0.5 +1.0 +1.5 E(V vs. S.C.E.) -400 Current density (A/m)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
Each volt amm ogram reaches a plateau at low potential beca...View the full answer

Answered By

Jemima Wangu
I am a proficient tutor with the following skills;
Effective time management
Microsoft Office
Computer skills
Multitasking skills
Strong Mathematical skills
Honesty and integrity
Advanced technology skills
Motivated attitude
Content writing
Content research and development
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The exchange current density for a PtIFe3+,Fel+ electrode is 2.5 mA cm-I. The standard potential of the electrode is +0.77 V. Calculate the current flowing through an electrode of surface area 1.0...
-
What is a density curve, and why are such curves important? In each of Problems 24, assume that the variable under consideration has a density curve. Note that the answers required here may be only...
-
Explain why species that overlap a great deal in their fundamental niches have a high probability of competing. Now explain why species that overlap a great deal in their realized niches and live in...
-
Holt Company purchased a computer for $8,000 on January 1, 2009. Straight-line depreciation is used, based on a 5-year life and a $1,000 salvage value . In 2011, the estimates are revised. Holt now...
-
What were the key elements in Blahna's successful diversity strategy?
-
Explain content negotiation using CC/PP. Draw a class diagram for the same.
-
Charles Smith, Inc., is a manufacturer of small office equipment. Smith transacts most of its business on credit and offers its customers credit terms of 2/10, n/30. On July 1, Smith shipped an order...
-
Antelope Manufacturing makes a component called A1030. This component is manufactured only when ordered by a customer, so Antelope keeps no inventory of A1030. The list price is $ 115 per unit, but...
-
The Lucky Business is looking at a project with the estimated cash flow as follows: Initial Investment at start of project: $ 3 , 6 0 0 , 0 0 0 Cash Flow at end of Year 1 : $ 5 0 0 , 0 0 0 Cash Flow...
-
Prepare A Balance Sheet And Income Statement For TNT, Inc., From The Scrambled List Of Items found here: a. Prepare an income statement for TNT. b. Prepare a balance sheet for TNT. c. What can you...
-
In Figure 17-11, 2.00 nmol of fructose were introduced at the time of the arrow. How many electrons are lost in the oxidation of one molecule of fructose? Compare the theoretical number of coulombs...
-
In 1 M NH 3/1 M NH 4 Cl solution, Cu 2+ is reduced to Cu+ near 20.3 V (versus S.C.E.), and Cu + is reduced to Cu(in Hg) near 20.6 V. (a) Sketch a qualitative sampled current polarogram for a solution...
-
There are two tangent lines from the point (0, 1) to the circle x + (y + 1) 2 = 1 (see figure). Find equations of these two lines by using the fact that each tangent line intersects the circle at...
-
Ladder Co. is expected to pay a dividend of $2.65 per share, one year from today. The company will increase its dividend by 10 percent per year for the following two years (dividends received two...
-
Your friend is tired of being single and wants to find someone with whom to have a relationship as soon as possible. Said friend, being inexperienced in relationships, asks you, "Who will be...
-
Jensen Sports has outstanding an issue of preferred stock with a par value of $100. It pays an annual dividend equal to 6% of par value. If the required return on Jensen preferred stock is 8%, and if...
-
Determine the total dividends and the per-share dividends declared on each class of stock for each of the six years. There were no dividends in arrears at the beginning of Year 1. Summarize the data...
-
People are constantly making first impressions in social, personal, and professional situations. What is a situation where you think it is especially important to make a positive first impression?...
-
State the empirical rule for variables?
-
The graph of an equation is given. (a) Find the intercepts. (b) Indicate whether the graph is symmetric with respect to the x-axis, the y-axis, or the origin. -3 6 -6 3 x
-
Calculate the pH of a solution prepared by mixing 0.0800 mol of chloroacetic acid plus 0.0400 mol of sodium chloroacetate in 1.00 L of water. (a) First do the calculation by assuming that the...
-
Calculate how many milliliters of 0.626 M KOH should be added to 5.00 g of MOBS (Table 8-2) to give a pH of 7.40.
-
(a) Use Equations 8-20 and 8-21 to find the pH and concentrations of HA and A- in a solution prepared by mixing 0.00200 mol of acetic acid plus 0.004 00 mol of sodium acetate in 1.00 L of water. (b)...
-
CNBC.com reported mortgage applications increased 9.9% due to a decrease in the rate on 30-year fixed-rate mortgages. Joe Sisneros wants to purchase a vacation home for $350,000 with 20% down....
-
Becky's company is considering an investment project. To start, Becky just needs to purchase an equipment priced at $5500. The equipment will follow a straight-line depreciation over 10 years. The...
-
10 10 1 point Refer to the adjustment grid below. -- Address Sale price Sale price/GLA Days on market Value adjustments Financing Concessions Date of sale/time 5.00% Location Leasehold/fee simple...

Study smarter with the SolutionInn App


