Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured
Question:
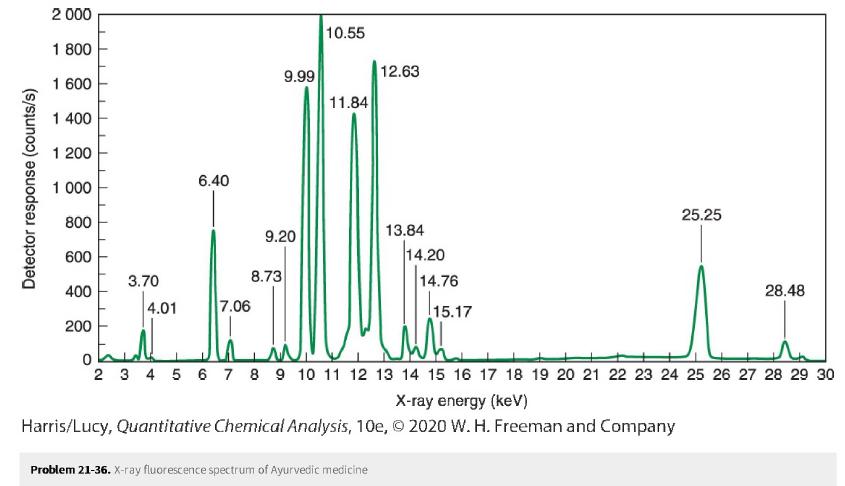
Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several toxic elements. In the spectrum below, identify as many peaks as you can in the X-ray fluorescence spectrum of the Ayurvedic medicine.
Transcribed Image Text:
Detector response (counts/s) 2 000 1 800 1 600 1 400 1 200 1 000 800 600 400 200 3.70 4.01 6.40 7.06 9.99 9.20 8.73 10.55 11.84 12.63 13.84 Problem 21-36. X-ray fluorescence spectrum of Ayurvedic medicine 14.20 14.76 15.17 X-ray energy (keV) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company 25.25 0 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 28.48
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Answer The peaks in the Xray fluorescence spectrum are 1 Cadmi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Holt Industries, Inc. does business in State 1. That state uses federal taxable income as the starting point for calculating state income tax. Interest on state obligations is taxed, but interest on...
-
1. Comment (briefly) on the design of the two costing systems + draw an overview diagram of each system. Explain why you believe / do not believe that the ABC system provides better costing...
-
Ayurveda is a form of medicine practiced in India. In one study, 20% of U.S.- and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several...
-
ABC Pty Ltd would like to set up a Virtualisation Platform on their organisation. You have been hired by Company to be their network and system administrator to implement virtualisation for...
-
Show that is as given in Eq. (11.2.5).
-
Discuss the major success factors for the Kuali Financials development effort?
-
Using the Internet, search for the terms "litiga- tion support" and "litigation services" and visit five or six sites that involve forensic accounting services. Also explore several sites involving...
-
Arrowbell Company is a growing company. Two years ago, it decided to expand in order to increase its production capacity. The company anticipates that the expansion program can be completed in...
-
PROBLEM: On January 1, 2021, JEFFORDS CORP. bought a piece of land worth Php 2,600,000. JEFFORDS' business model is to buy and sell the land. On February 1, 2024, JEFFORDS decided to lease the land...
-
Jerry Smith is thinking about opening a bicycle shop in his hometown. Jerry loves to take his own bike on 50-mile trips with his friends, but he believes that any small business should be started...
-
Signal in attenuated total reflectance is related to penetration depth. a. Calculate the penetration depth of 1 500 cm -1 1500 cm -1 radiation into an organic sample (n2=1.5)(n 2 =1.5) for a 45450...
-
a. Explain the purpose of the collision cell in Figure 21-28. b. Determination of 28Si 28Si , < experiences isobaric interference from 14N2+ 14N + 2 and background silicon leached from glass...
-
ATP is a (+) allosteric effector, and CTP is a (-) allosteric effector of the enzyme ATCase. Both of these heterotropic effectors bind to the regulatory subunits on ATCase. The substrates of ATCase,...
-
Wally is pleased with your work. He asks you for help on one more project - and then you can take a well-deserved break! He gives you this information that he has collected on one of IanCo's key...
-
If your car payment is $1250 per month, your minimum credit card payment is $1050 per month, your mortgage is $4400 per month, and your after tax monthly income is $6400, what is your debt payment...
-
Consider two charges Q = 100 C and Q = -200 C as shown in the picture below. a. Determine magnitude and direction of the net electric field at point A. b. Determine the magnitude and direction of the...
-
Johnny Rockefeller had a bad credit rating and went to a local cash center. He took out a $100.00 loan payable intwo weeks for $105.00. What is the percent of interest paid on this loan?
-
Based upon these amounts, what is total stockholders' equity at the end of the current year? Beginning balance in retained earnings: $34,569 Net income during the year of $15,960 Dividends declared...
-
Pavin acquires all of Stabler's outstanding shares on January 1, 2011, for $460,000 in cash. Of this amount, $30,000 was attributed to equipment with a 10-year remaining life and $40,000 was assigned...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
The cumulative formation constant for SnCl2(aq) in 1.0 M NaNO3 is 2 = 12. Find the concentration of SnCl2(aq) for a solution in which the concentrations of Sn2+ and Cl- are both somehow fixed at 0.20...
-
Given the following equilibria, calculate the concentration of each zinc species in a solution saturated with Zn(OH)2(s) and containing [OH-] at a fixed concentration of 3.2 10-7M. Zn(OH)2(s) Ksp =...
-
Although KOH, RbOH, and CsOH have little association between metal and hydroxide in aqueous solution, Li+ and Na+ doform complexes with OH-: Na + + OH- NaoH (aq) K1 = 0.20 Prepare a table like the...
-
2. If a company's assets are $100,000, liabilities are $50,000, and equity is $50,000, what is the company's debt-to- equity ratio? a) 0.5 b) 1 c) 2 d) 1.5 3. Which accounting principle requires that...
-
A river habitat rehabilitation is being considered. If a BCA is to be undertaken which or the following should be part of/included in the accounting stance? Group of answer choices a discount rate...
-
about the burger king one risk in each of the 4 ERM risk categories. Out of those risks you will choose one to do a risk assessment on as explained in Chapter 3. And finally, you will describe how...

Study smarter with the SolutionInn App


