Blank solutions were monitored for vanadium as 51V+ 51 V + using inductively coupled plasma-mass spectrometry with
Question:
Blank solutions were monitored for vanadium as 51V+ 51V+ using inductively coupled plasma-mass spectrometry with the following results.
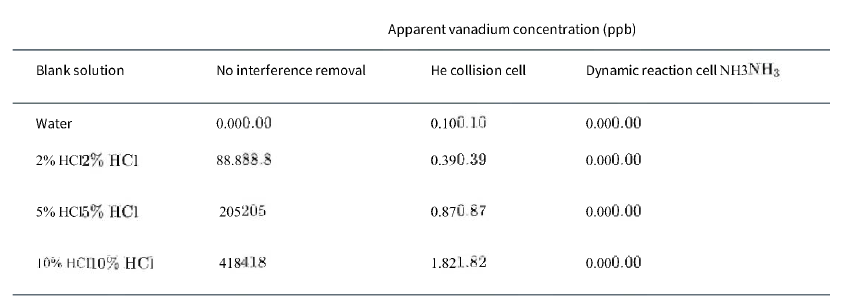
a. Is the HClHCl used to prepare the blank solutions contaminated with vanadium?
b. Would monitoring 50V+50V+ be a better alternative?
c. What acid should be used in place of HClHCl?
Transcribed Image Text:
Blank solution Water 2% HC12% HC1 5% HCI5% HC1 10% HC110% HCI No interference removal 0.000.00 88.888.8 205205 418418 Apparent vanadium concentration (ppb) He collision cell 0.100.10 0.390.39 0.870.87 1.821.82 Dynamic reaction cell NH3NH3 0.000.00 0.000.00 0.000.00 0.000.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Answer a No the HClHCl used to prepare the blank s...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Harold conducts a business with the following results for the year: Revenue ......................................................... $20,000 Depreciation on car...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
What nonparametric test can be used to compare plasma aldosterone group between Caucasian and African American children? Hypertension Plasma aldosterone has been related to blood-pressure levels in...
-
Starting on January 1, 2024, you put $125 every month into an account with an APR of 4.10%. Your last payment is on December 1, 2032. On January 1 of that year, the balance in the account, rounded to...
-
Moore and McCabe (1999, p. 174) report prices paid for several species of seafood in 1970 and 1980. These values are in Table 11.6. If we were interested in trying to predict 1980 seafood prices from...
-
Why did you propose that Benton hire a consultant to help choose a vendor?
-
An AICPA Fraud Task Force suggests that if a forensic accountant is brought into an audit to conduct a separate investigation (and not an adjunct to an audit), what standards apply?
-
Techno Designs Store uses the accounts receivable aging method to estimate uncollectible accounts. On February 1, 2014, the balance of the Accounts Receivable account was a debit of $442,341, and the...
-
1 Take Test: FinalExamTst blackboard.olemiss.edu In SAP when recognizing the receipt of goods, O A. a manual entry must be made in both the material management module and the financial accounting...
-
The following on-boarding process for new employees has two stages. In the first, employees fill out several government forms and necessary visas for immigration. In the second the employees attend...
-
Calibration curve. Figure 21-30 describes graphite furnace atomic absorbance determination of silver within individual Daphnia magna. Calibration data at 328.068 nn 328.068 nn is shown below. a....
-
What is the difference between luminescence, chemiluminescence, and bioluminescence?
-
LN Consulting is a calendar year, cash basis unincorporated business. The business is not required to provide audited financial statements to any external user. LN's accounting records show the...
-
The figure shows a circuit with four capacitors and a voltage source of V = 19 V. The capacitances are: C1 = 11 nF, C2 = 7 nF, C3 = 8 nF, and C4 = 10 nF. Find the voltage across capacitor 3. C 2 C3...
-
The Ontario and Canadian governments had announced reducing fees for regulated child care by 50% on average by the end of 2022 and.bringing fees for regulated child care down to $10-a-day on average...
-
How does a higher EPS potentially impact a company's stock price? 8. What are the limitations of using EPS as a standalone measure of a company's financial performance? 9. How can investors compare...
-
The net income of Amazon decreased from $0.63 billion in 2011 to -$0.24 billion in 2014. a. Use this information to find a linear model for Amazon's net income N (in billions of dollars) as a...
-
Consider that consumer price acceptance is not driven by value as is assumed in value-based pricing. This would imply that people only pay for the product features. In fact, people pay for much more...
-
Through the payment of $10,468,000 in cash, Drexel Company acquires voting control over Young Company. This price is paid for 60 percent of the subsidiary's 100,000 outstanding common shares ($40 par...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Use Table 6-1 to calculate the pH of pure water at (a) 25C and (b) 100C.
-
An acidic solution containing 0.010 M La3+ is treated with NaOH until La(OH)3 precipitates. At what pH does this occur?
-
Use Le Chtelier's principle and Kw in Table 6-1 to decide whether the autoprotolysis of water is endothermic or exothermic at (a) 25C; (b) 100C; (c) 300C.
-
Possible new mines in Arizona include a proposed new (and potentially very large) underground mine just east of the smaller town of _____ and near the Oak Flat campground. Group of answer choices...
-
Fill in the Blank Question Fill in the blank question. power is the power of knowledge
-
Harold Lloyd learned slapstick on the vaudeville circuit. Group of answer choices True False

Study smarter with the SolutionInn App


