Consider the cyclic voltammogram of the Co 3+ compound Co(B 9 C 2 H 11 )2. Suggest
Question:
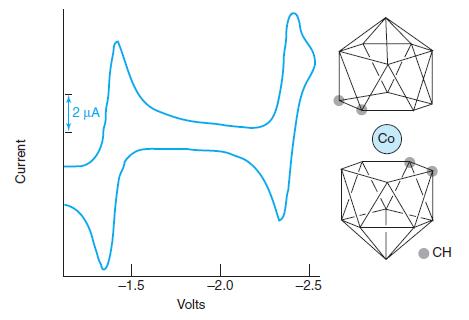
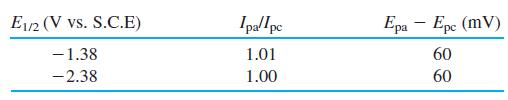
Consider the cyclic voltammogram of the Co3+ compound Co(B9C2H11)2̅. Suggest a chemical reaction to account for each wave. Are the reactions reversible? How many electrons are involved in each step? Sketch the sampled current and square wave polarograms expected for this compound.
Transcribed Image Text:
2 µA Co CH -1.5 -2.0 -2.5 Volts Current
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
A cyclic voltammogram of the Co3 compound CoB9C2H112 provides information about the electrochemical ...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider the following energy profile (a) How many elementary reactions are in the reaction mechanism? (b) How many intermediates are formed in the reaction? (c) Which step is rate limiting? (d) Is...
-
For each of the following chemical reactions, write the correct element and/or compound symbols, formulas, and coefficients needed to produce complete, balanced equations. In all cases, the reactants...
-
A catalyst affects which one or more of the following? a. the energy needed for a chemical reaction to occur b. the energy a chemical reaction gives off c. the speed of a chemical reaction d. whether...
-
Your geothermal heat pump is used to heat your house in the winter. The heat pump is operated as a vapor-compression cycle. The design is such that there must be a 10 degree temperature difference...
-
Last week's total number of hours worked by a student, y, depends on the number of days, x, he reported to work last week. Suppose the data from nine students provided (a) Plot the scatter diagram....
-
For the graph below, write the adjacency matrix \(A\), compute \(A^{3}\), and verify that for each \(i\) and \(j, A^{3}(i, j)\) is the number of paths from \(i\) to \(j\) of length 3 by listing those...
-
\(72 \div 6+18\) Perform the indicated calculation.
-
Hearty Soup Co. uses a process cost system to record the costs of processing soup, which requires the cooking and filling processes. Materials are entered from the cooking process at the beginning of...
-
. On 21" Feb, you sold 800 shares of Ace stock for $24,000. The sale was a short sale with an initial margin requirement of 70 percent. The maintenance margin is 40 percent. On 23"d Feb, s...
-
The donkey driver Donkey King is using a donkey to carry goods from Harbor City to a village up in the mountains. The people living in the village are willing to buy gold, silk, salt, and sponge....
-
A type of instruction that produce several lines of machine language code is a (A) Assemble (B) Address (C) Mnemonic (D) Macro
-
In a coulometric Karl Fischer water analysis, 25.00 mL of pure dry methanol required 4.23 C to generate enough I 2 to react with the residual H 2 O in the methanol. A suspension of 0.847 6 g of...
-
Of the 19 L amino acids, 18 have the S configuration at the carbon. Cysteine is the only L amino acid that has an R configuration. Explain.
-
What type of management process function is being used when a manager motivates the workforce by rewarding them with additional vacation when standards are achieved?
-
A thermometer with an initial temperature of 65 F is placed in an oven heated to a constant temperature. The thermometer reads 125 F after 1 minute and 170 F after 2 minutes. What is the temperature...
-
Successful career means getting multiple jobs, promotions and earning a handsome salary. Discuss whether you agree with the statement
-
To estimate the difference between the average kilometres per litre of two different models of car, samples are taken and the following information is collected. (10p+5p) Sample size Sample mean...
-
For this circuit, use the node numbers indicated in the figure; show your work in detail. 1 20 www 5A () www 30: 514 14292 Vout Output Port Solve the circuit and find the output voltage. Find Req...
-
Who might participate in the social media interaction on behalf of a company? What strength could each of these people contribute to the conversation?
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
Using the linear calibration curve in Figure 4-13, find the quantity of unknown protein that gives a measured absorbance of 0.264 when a blank has an absorbance of 0.095. Figure 4-13 0.40 Unknown...
-
Consider the least-squares problem in Figure 4-11. (a) Suppose that a single new measurement produces a y value of 2.58. Find the corresponding x value and its uncertainty. (b) Suppose you measure y...
-
Consider the linear calibration curve in Figure 4-13, which is derived from the 14 corrected absorbances in the shaded region at the right side of Table 4-7. Create a least-squares spreadsheet like...
-
The judgment creditor may have to issue _______________, or questions to banks and other financial institutions, in order to locate assets. Group of answer choices Interrogatories in aid of...
-
nyone analyzing a firm's financial statements should: Group of answer choices All of these answers perform a benchmark analysis do a trend analysis use audited financial statements
-
The direct exchange of one good for another without the use of money is Multiple choice question. marketplace open trade barter market transaction

Study smarter with the SolutionInn App


